Exogenous Putrescine Modulates Nitrate Reductase-Dependent NO Production in Cucumber Seedlings Subjected to Salt Stress
- PMID: 37755310
- PMCID: PMC10535175
- DOI: 10.3390/metabo13091030
Exogenous Putrescine Modulates Nitrate Reductase-Dependent NO Production in Cucumber Seedlings Subjected to Salt Stress
Abstract
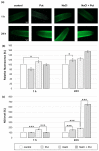
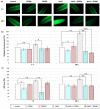
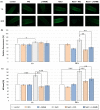
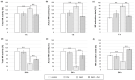
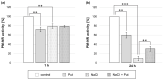
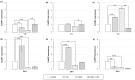
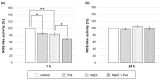
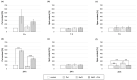
Polyamines (PAs) are small aliphatic compounds that participate in the plant response to abiotic stresses. They also participate in nitric oxide (NO) production in plants; however, their role in this process remains unknown. Therefore, the study aimed to investigate the role of putrescine (Put) in NO production in the roots of cucumber seedlings subjected to salt stress (120 mM NaCl) for 1 and 24 h. In salinity, exogenous Put can regulate NO levels by managing NO biosynthesis pathways in a time-dependent manner. In cucumber roots exposed to 1 h of salinity, exogenous Put reduced NO level by decreasing nitrate reductase (NR)-dependent NO production and reduced nitric oxide synthase-like (NOS-like) activity. In contrast, during a 24 h salinity exposure, Put treatment boosted NO levels, counteracting the inhibitory effect of salinity on the NR and plasma membrane nitrate reductase (PM-NR) activity in cucumber roots. The role of endogenous Put in salt-induced NO generation was confirmed using Put biosynthesis inhibitors. Furthermore, the application of Put can modulate the NR activity at the genetic and post-translational levels. After 1 h of salt stress, exogenous Put upregulated CsNR1 and CsNR2 expression and downregulated CsNR3 expression. Put also decreased the NR activation state, indicating a reduction in the level of active dephosphorylated NR (dpNR) in the total enzyme pool. Conversely, in the roots of plants subjected to 24 h of salinity, exogenous Put enhanced the NR activation state, indicating an enhancement of the dpNR form in the total NR pool. These changes were accompanied by a modification of endogenous PA content. Application of exogenous Put led to an increase in the amount of Put in the roots and reduced endogenous spermine (Spm) content in cucumber roots under 24 h salinity. The regulatory role of exogenous Put on NO biosynthesis pathways may link with plant mechanisms of response to salt stress.
Keywords: nitrate reductase (NR); nitric oxide; nitric oxide synthase-like activity; polyamines; putrescine; salt stress.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Butcher K., Wick A.F., DeSutter T., Chatterjee A., Harmon J. Soil Salinity: A Threat to Global Food Security. Agron. J. 2016;108:2189–2200. doi: 10.2134/agronj2016.06.0368. - DOI
-
- Hasanuzzaman M., Oku H., Nahar K., Bhuyan M.H.M.B., Mahmud J.A., Baluska F., Fujita M. Nitric Oxide-Induced Salt Stress Tolerance in Plants: ROS Metabolism, Signaling, and Molecular Interactions. Plant Biotechnol. Rep. 2018;12:77–92. doi: 10.1007/s11816-018-0480-0. - DOI
-
- Nahar K., Hasanuzzaman M., Rahman A., Alam M.M., Mahmud J.-A., Suzuki T., Fujita M. Polyamines Confer Salt Tolerance in Mung Bean (Vigna radiata L.) by Reducing Sodium Uptake, Improving Nutrient Homeostasis, Antioxidant Defense, and Methylglyoxal Detoxification Systems. Front. Plant Sci. 2016;7:1104. doi: 10.3389/fpls.2016.01104. - DOI - PMC - PubMed
-
- Rasool S., Hameed A., Azooz M.M., Muneeb-u-Rehman, Siddiqi T.O., Ahmad P. Salt Stress: Causes, Types and Responses of Plants. In: Ahmad P., Azooz M.M., Prasad M.N.V., editors. Ecophysiology and Responses of Plants under Salt Stress. Springer; New York, NY, USA: 2013. pp. 1–24.
LinkOut - more resources
Full Text Sources

