Increased DNMT1 Involvement in the Activation of LO2 Cell Death Induced by Silver Nanoparticles via Promoting TFEB-Dependent Autophagy
- PMID: 37755761
- PMCID: PMC10537645
- DOI: 10.3390/toxics11090751
Increased DNMT1 Involvement in the Activation of LO2 Cell Death Induced by Silver Nanoparticles via Promoting TFEB-Dependent Autophagy
Abstract
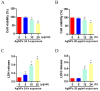
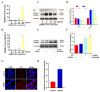
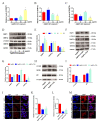
The accumulation of exogenous silver nanoparticles (AgNPs) will terminally bring about liver injury, including cell death, where DNA methylation tends to be a crucial epigenetic modulator. The change in the cell autophagy level verified to be closely associated with hepatocyte death has been followed with wide interest. But the molecular toxicological mechanisms of AgNPs in relation to DNA methylation, autophagy, and cell death remain inconclusive. To address the issue above, in LO2 cells treated with increasing concentrations of AgNPs (0, 5, 10, and 20 μg/mL), a cell cytotoxicity assay was performed to analyze the level of cell death, which also helped to choose an optimal concentration for next experiments. An immunofluorescence assay was used to determine the autophagic flux as well as TFEB translocation, with qRT-PCR and western blot being used to analyze the expression level of autophagy-related genes and proteins. According to our findings, in the determination of cell viability, 20 μg/mL (AgNPs) was adopted as the best working concentration. LO2 cell death, autophagy, and TFEB nuclear translocation were induced by AgNPs, which could be inhibited by lysosome inhibitor chloroquine (CQ) or siRNA specific for TFEB. Moreover, AgNP exposure led to DNA hypermethylation, with DNMT1 taking part mainly, which could be obviously prevented by 5-Aza-2'-deoxycytidine (5-AzaC) or trichostatin A (TSA) treatment or DNMT1 knockout in LO2 cells. Our studies suggest that through TFEB-dependent cell autophagy, increased DNMT1 may facilitate cell death induced by AgNPs.
Keywords: DNMT1; autophagy; cell death; hepatocyte injury; silver nanoparticles.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as potential conflicts of interest.
Figures


Similar articles
-
Silver nanoparticles induce lysosomal-autophagic defects and decreased expression of transcription factor EB in A549 human lung adenocarcinoma cells.Toxicol In Vitro. 2018 Feb;46:148-154. doi: 10.1016/j.tiv.2017.10.009. Epub 2017 Oct 5. Toxicol In Vitro. 2018. PMID: 28987793
-
Functional Autophagic Flux Regulates AgNP Uptake And The Internalized Nanoparticles Determine Tumor Cell Fate By Temporally Regulating Flux.Int J Nanomedicine. 2019 Nov 20;14:9063-9076. doi: 10.2147/IJN.S222211. eCollection 2019. Int J Nanomedicine. 2019. PMID: 31819419 Free PMC article.
-
Silica Nanoparticles Trigger Chaperone HSPB8-Assisted Selective Autophagy via TFEB Activation in Hepatocytes.Small. 2023 Feb;19(5):e2204310. doi: 10.1002/smll.202204310. Epub 2022 Dec 4. Small. 2023. PMID: 36464658
-
Silver nanoparticles induce protective autophagy via Ca2+/CaMKKβ/AMPK/mTOR pathway in SH-SY5Y cells and rat brains.Nanotoxicology. 2019 Apr;13(3):369-391. doi: 10.1080/17435390.2018.1550226. Epub 2019 Feb 7. Nanotoxicology. 2019. PMID: 30729847
-
Mechanisms of silver nanoparticle-induced toxicity and important role of autophagy.Nanotoxicology. 2016 Oct;10(8):1021-40. doi: 10.1080/17435390.2016.1189614. Epub 2016 Jun 20. Nanotoxicology. 2016. PMID: 27240148 Review.
Cited by
-
Impaired TFEB-mediated autophagy-lysosome fusion promotes tubular cell cycle G2/M arrest and renal fibrosis by suppressing ATP6V0C expression and interacting with SNAREs.Int J Biol Sci. 2024 Mar 3;20(5):1905-1926. doi: 10.7150/ijbs.91480. eCollection 2024. Int J Biol Sci. 2024. PMID: 38481802 Free PMC article.
References
Grants and funding
- 2018A0303130240; 2020A151501204; 2020A1515110614; 2021B1515140032/Natural Science Foundation of Guangdong Province
- 82103879/National Natural Science Foundation of China
- B2019027; GDMUB2020013/Scientific Research Funding of Guangdong Medical University
- A2018225; A2020189/the Medical Scientific Research Funding of Guangdong Province
- 4SG22021G; 4SG22260G; 4SG23001G/Discipline construction project of Guangdong Medical University
LinkOut - more resources
Full Text Sources
Research Materials

