Estrogen receptor beta in astrocytes modulates cognitive function in mid-age female mice
- PMID: 37758709
- PMCID: PMC10533869
- DOI: 10.1038/s41467-023-41723-7
Estrogen receptor beta in astrocytes modulates cognitive function in mid-age female mice
Abstract
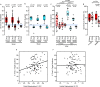
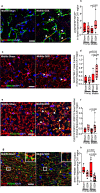
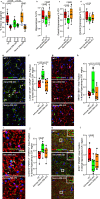
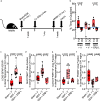
Menopause is associated with cognitive deficits and brain atrophy, but the brain region and cell-specific mechanisms are not fully understood. Here, we identify a sex hormone by age interaction whereby loss of ovarian hormones in female mice at midlife, but not young age, induced hippocampal-dependent cognitive impairment, dorsal hippocampal atrophy, and astrocyte and microglia activation with synaptic loss. Selective deletion of estrogen receptor beta (ERβ) in astrocytes, but not neurons, in gonadally intact female mice induced the same brain effects. RNA sequencing and pathway analyses of gene expression in hippocampal astrocytes from midlife female astrocyte-ERβ conditional knock out (cKO) mice revealed Gluconeogenesis I and Glycolysis I as the most differentially expressed pathways. Enolase 1 gene expression was increased in hippocampi from both astrocyte-ERβ cKO female mice at midlife and from postmenopausal women. Gain of function studies showed that ERβ ligand treatment of midlife female mice reversed dorsal hippocampal neuropathology.
© 2023. Springer Nature Limited.
Conflict of interest statement
The authors had full access to all of the data in this study and take complete responsibility for the integrity of the data and the accuracy of the data analysis. R.R.V. is an inventor on UCLA’s patents pertaining to estriol and is a consultant with equity in CleopatraRX, the licensee. R.R.V. is also an inventor on UCLA’s patents pertaining to novel estrogen receptor beta ligands which are unlicensed. Other authors have nothing to disclose.
Figures