Coupled Electrostatic and Hydrophobic Destabilisation of the Gelsolin-Actin Complex Enables Facile Detection of Ovarian Cancer Biomarker Lysophosphatidic Acid
- PMID: 37759826
- PMCID: PMC10527313
- DOI: 10.3390/biom13091426
Coupled Electrostatic and Hydrophobic Destabilisation of the Gelsolin-Actin Complex Enables Facile Detection of Ovarian Cancer Biomarker Lysophosphatidic Acid
Abstract
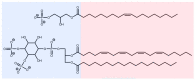
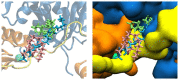
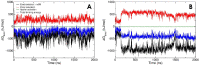
Lysophosphatidic acid (LPA) is a promising biomarker candidate to screen for ovarian cancer (OC) and potentially stratify and treat patients according to disease stage. LPA is known to target the actin-binding protein gelsolin which is a key regulator of actin filament assembly. Previous studies have shown that the phosphate headgroup of LPA alone is inadequate to bind to the short chain of amino acids in gelsolin known as the PIP2-binding domain. Thus, the molecular-level detail of the mechanism of LPA binding is poorly understood. Here, we model LPA binding to the PIP2-binding domain of gelsolin in the gelsolin-actin complex through extensive ten-microsecond atomistic molecular dynamics (MD) simulations. We predict that LPA binding causes a local conformational rearrangement due to LPA interactions with both gelsolin and actin residues. These conformational changes are a result of the amphipathic nature of LPA, where the anionic phosphate, polar glycerol and ester groups, and lipophilic aliphatic tail mediate LPA binding via charged electrostatic, hydrogen bonding, and van der Waals interactions. The negatively-charged LPA headgroup binds to the PIP2-binding domain of gelsolin-actin while its hydrophobic tail is inserted into actin, creating a strong LPA-insertion pocket that weakens the gelsolin-actin interface. The computed structure, dynamics, and energetics of the ternary gelsolin-LPA-actin complex confirms that a quantitative OC assay is possible based on LPA-triggered actin release from the gelsolin-actin complex.
Keywords: actin; gelsolin; lipid-protein interaction; lysophosphatidic acid; ovarian cancer; predictive molecular modelling.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
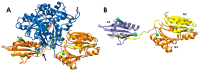


Similar articles
-
Design and Characterization of a Dual-Protein Strategy for an Early-Stage Assay of Ovarian Cancer Biomarker Lysophosphatidic Acid.Biosensors (Basel). 2024 Jun 2;14(6):287. doi: 10.3390/bios14060287. Biosensors (Basel). 2024. PMID: 38920591 Free PMC article.
-
Gelsolin and functionally similar actin-binding proteins are regulated by lysophosphatidic acid.EMBO J. 1998 Oct 15;17(20):5923-32. doi: 10.1093/emboj/17.20.5923. EMBO J. 1998. PMID: 9774337 Free PMC article.
-
Gelsolin binding and cellular presentation of lysophosphatidic acid.J Biol Chem. 2000 May 12;275(19):14573-8. doi: 10.1074/jbc.275.19.14573. J Biol Chem. 2000. PMID: 10799543
-
[Lysophosphatidic acid as a potential target for treatment and molecular diagnosis of epithelial ovarian cancers].Orv Hetil. 2009 Jun 14;150(24):1109-18. doi: 10.1556/oh.2009.28631. Orv Hetil. 2009. PMID: 19739275 Review. Hungarian.
-
Critical role of lysophospholipids in the pathophysiology, diagnosis, and management of ovarian cancer.Cancer Treat Res. 2002;107:259-83. doi: 10.1007/978-1-4757-3587-1_12. Cancer Treat Res. 2002. PMID: 11775454 Review.
Cited by
-
Acoustic Wave Sensor Detection of an Ovarian Cancer Biomarker with Antifouling Surface Chemistry.Sensors (Basel). 2024 Dec 10;24(24):7884. doi: 10.3390/s24247884. Sensors (Basel). 2024. PMID: 39771624 Free PMC article.
-
The Emerging Role of LPA as an Oncometabolite.Cells. 2024 Apr 4;13(7):629. doi: 10.3390/cells13070629. Cells. 2024. PMID: 38607068 Free PMC article. Review.
-
Design and Characterization of a Dual-Protein Strategy for an Early-Stage Assay of Ovarian Cancer Biomarker Lysophosphatidic Acid.Biosensors (Basel). 2024 Jun 2;14(6):287. doi: 10.3390/bios14060287. Biosensors (Basel). 2024. PMID: 38920591 Free PMC article.
References
-
- Tate A.R., Martin A.G.R., Murray-Thomas T., Anderson S.R., Cassell J.A. Determining the Date of Diagnosis—Is It a Simple Matter? The Impact of Different Approaches to Dating Diagnosis on Estimates of Delayed Care for Ovarian Cancer in UK Primary Care. BMC Med. Res. Methodol. 2009;9:42. doi: 10.1186/1471-2288-9-42. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

