Enhanced Expansion of Human Pluripotent Stem Cells and Somatic Cell Reprogramming Using Defined and Xeno-Free Culture Conditions
- PMID: 37760101
- PMCID: PMC10525589
- DOI: 10.3390/bioengineering10090999
Enhanced Expansion of Human Pluripotent Stem Cells and Somatic Cell Reprogramming Using Defined and Xeno-Free Culture Conditions
Abstract
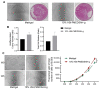
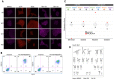
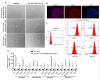
Human embryonic stem cells and induced pluripotent stem cells (hPSC) have an unprecedented opportunity to revolutionize the fields of developmental biology as well as tissue engineering and regenerative medicine. However, their applications have been significantly limited by the lack of chemically defined and xeno-free culture conditions. The demand for the high-quality and scaled-up production of cells for use in both research and clinical studies underscores the need to develop tools that will simplify the in vitro culture process while reducing the variables. Here, we describe a systematic study to identify the optimal conditions for the initial cell attachment of hPSC to tissue culture dishes grafted with polymers of N-(3-Sulfopropyl)-N-Methacryloxyethyl-N, N-Dimethylammoniun Betaine (PMEDSAH) in combination with chemically defined and xeno-free culture media. After testing multiple supplements and chemicals, we identified that pre-conditioning of PMEDSAH grafted plates with 10% human serum (HS) supported the initial cell attachment, which allowed for the long-term culture and maintenance of hPSC compared to cells cultured on Matrigel-coated plates. Using this culture condition, a 2.1-fold increase in the expansion of hPSC was observed without chromosomal abnormalities. Furthermore, this culture condition supported a higher reprogramming efficiency (0.37% vs. 0.22%; p < 0.0068) of somatic cells into induced pluripotent stem cells compared to the non-defined culture conditions. This defined and xeno-free hPSC culture condition may be used in obtaining the large populations of hPSC and patient-derived iPSC required for many applications in regenerative and translational medicine.
Keywords: PMEDSAH; human embryonic stem cells; human pluripotent stem cells; reprogramming; self-renewal; synthetic polymer grafting.
Conflict of interest statement
LVD declares a conflict of interest.
Figures
Similar articles
-
Discovery of a Novel Polymer for Xeno-Free, Long-Term Culture of Human Pluripotent Stem Cell Expansion.Adv Healthc Mater. 2021 Mar;10(6):e2001448. doi: 10.1002/adhm.202001448. Epub 2020 Dec 28. Adv Healthc Mater. 2021. PMID: 33369242 Free PMC article.
-
A defined and xeno-free culture method enabling the establishment of clinical-grade human embryonic, induced pluripotent and adipose stem cells.PLoS One. 2010 Apr 19;5(4):e10246. doi: 10.1371/journal.pone.0010246. PLoS One. 2010. PMID: 20419109 Free PMC article.
-
A defined xeno-free and feeder-free culture system for the derivation, expansion and direct differentiation of transgene-free patient-specific induced pluripotent stem cells.Biomaterials. 2014 Mar;35(9):2816-26. doi: 10.1016/j.biomaterials.2013.12.050. Epub 2014 Jan 9. Biomaterials. 2014. PMID: 24411336
-
Production of human pluripotent stem cell therapeutics under defined xeno-free conditions: progress and challenges.Stem Cell Rev Rep. 2015 Feb;11(1):96-109. doi: 10.1007/s12015-014-9544-x. Stem Cell Rev Rep. 2015. PMID: 25077810 Free PMC article. Review.
-
Microcarrier-based platforms for in vitro expansion and differentiation of human pluripotent stem cells in bioreactor culture systems.J Biotechnol. 2016 Sep 20;234:71-82. doi: 10.1016/j.jbiotec.2016.07.023. Epub 2016 Jul 29. J Biotechnol. 2016. PMID: 27480342 Review.
References
-
- López S.P., Hernández J.O. Advances in stem cell therapy. Adv. Exp. Med. Biol. 2012;741:290–313. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

