Repeated Transcranial Photobiomodulation with Light-Emitting Diodes Improves Psychomotor Vigilance and EEG Networks of the Human Brain
- PMID: 37760145
- PMCID: PMC10525861
- DOI: 10.3390/bioengineering10091043
Repeated Transcranial Photobiomodulation with Light-Emitting Diodes Improves Psychomotor Vigilance and EEG Networks of the Human Brain
Abstract
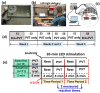
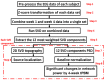
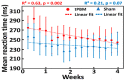
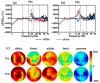
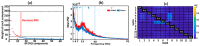
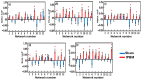
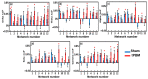
Transcranial photobiomodulation (tPBM) has been suggested as a non-invasive neuromodulation tool. The repetitive administration of light-emitting diode (LED)-based tPBM for several weeks significantly improves human cognition. To understand the electrophysiological effects of LED-tPBM on the human brain, we investigated alterations by repeated tPBM in vigilance performance and brain networks using electroencephalography (EEG) in healthy participants. Active and sham LED-based tPBM were administered to the right forehead of young participants twice a week for four weeks. The participants performed a psychomotor vigilance task (PVT) during each tPBM/sham experiment. A 64-electrode EEG system recorded electrophysiological signals from each participant during the first and last visits in a 4-week study. Topographical maps of the EEG power enhanced by tPBM were statistically compared for the repeated tPBM effect. A new data processing framework combining the group's singular value decomposition (gSVD) with eLORETA was implemented to identify EEG brain networks. The reaction time of the PVT in the tPBM-treated group was significantly improved over four weeks compared to that in the sham group. We observed acute increases in EEG delta and alpha powers during a 10 min LED-tPBM while the participants performed the PVT task. We also found that the theta, beta, and gamma EEG powers significantly increased overall after four weeks of LED-tPBM. Combining gSVD with eLORETA enabled us to identify EEG brain networks and the corresponding network power changes by repeated 4-week tPBM. This study clearly demonstrated that a 4-week prefrontal LED-tPBM can neuromodulate several key EEG networks, implying a possible causal effect between modulated brain networks and improved psychomotor vigilance outcomes.
Keywords: EEG; PVT; electroencephalography; light-emitting diodes; psychomotor vigilance task; repeated tPBM; tPBM; transcranial photobiomodulation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Combination of Group Singular Value Decomposition and eLORETA Identifies Human EEG Networks and Responses to Transcranial Photobiomodulation.Front Hum Neurosci. 2022 May 10;16:853909. doi: 10.3389/fnhum.2022.853909. eCollection 2022. Front Hum Neurosci. 2022. PMID: 35620152 Free PMC article.
-
Site- and electroencephalogram-frequency-specific effects of 800-nm prefrontal transcranial photobiomodulation on electroencephalogram global network topology in young adults.Neurophotonics. 2025 Jan;12(1):015011. doi: 10.1117/1.NPh.12.1.015011. Epub 2025 Feb 27. Neurophotonics. 2025. PMID: 40018415 Free PMC article.
-
Pulsed transcranial photobiomodulation generates distinct beneficial neurocognitive effects compared with continuous wave transcranial light.Lasers Med Sci. 2023 Sep 5;38(1):203. doi: 10.1007/s10103-023-03865-4. Lasers Med Sci. 2023. PMID: 37668791 Clinical Trial.
-
Transcranial photobiomodulation in the management of brain disorders.J Photochem Photobiol B. 2021 Aug;221:112207. doi: 10.1016/j.jphotobiol.2021.112207. Epub 2021 May 5. J Photochem Photobiol B. 2021. PMID: 34119804
-
Transcranial, Red/Near-Infrared Light-Emitting Diode Therapy to Improve Cognition in Chronic Traumatic Brain Injury.Photomed Laser Surg. 2016 Dec;34(12):610-626. doi: 10.1089/pho.2015.4037. Photomed Laser Surg. 2016. PMID: 28001756 Review.
Cited by
-
Modifying Alzheimer's disease pathophysiology with photobiomodulation: model, evidence, and future with EEG-guided intervention.Front Neurol. 2024 Aug 23;15:1407785. doi: 10.3389/fneur.2024.1407785. eCollection 2024. Front Neurol. 2024. PMID: 39246604 Free PMC article. Review.
-
Directed physiological networks in the human prefrontal cortex at rest and post transcranial photobiomodulation.Sci Rep. 2024 May 3;14(1):10242. doi: 10.1038/s41598-024-59879-7. Sci Rep. 2024. PMID: 38702415 Free PMC article.
-
Directed physiological networks in the human prefrontal cortex at rest and post transcranial photobiomodulation.Res Sq [Preprint]. 2023 Oct 6:rs.3.rs-3393702. doi: 10.21203/rs.3.rs-3393702/v1. Res Sq. 2023. Update in: Sci Rep. 2024 May 3;14(1):10242. doi: 10.1038/s41598-024-59879-7. PMID: 37886539 Free PMC article. Updated. Preprint.
References
-
- Hamblin M.R. Photobiomodulation in the Brain. Academic Press; Cambridge, MA, USA: 2019. Mechanisms of photobiomodulaion in the brain.
Grants and funding
LinkOut - more resources
Full Text Sources

