Examining the Effects of Hyperbaric Oxygen Therapy on the Cardiovascular System and Oxidative Stress in Insulin-Treated and Non-Treated Diabetic Rats
- PMID: 37760247
- PMCID: PMC10525412
- DOI: 10.3390/ani13182847
Examining the Effects of Hyperbaric Oxygen Therapy on the Cardiovascular System and Oxidative Stress in Insulin-Treated and Non-Treated Diabetic Rats
Abstract
Background: This study explored the effects of hyperbaric oxygen therapy (HBOT) on the cardiovascular system and oxidative stress in streptozotocin-induced diabetic rats. Wistar albino rats were divided into four groups: DM group (diabetic rats), DM+HBOT group (diabetic rats exposed to HBOT for 1 h daily, five days a week, at 2.8 atmosphere absolute (ATA) with 100% oxygen for two weeks), DM+INS group (diabetic rats treated with neutral protamine hagedorn (NPH) insulin at a dosage of 3-5 U/day), and DM+HBOT+INS group (diabetic rats treated with both NPH insulin and HBOT for two weeks).
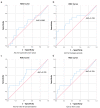
Methods: Evaluations included glycemic control, oxidative stress parameters, and cardiac function measurements.
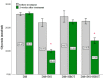
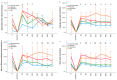
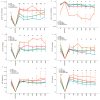
Results: NPH insulin treatment reduced blood glucose levels, although normoglycemia was not achieved. The DM+HBOT+INS group demonstrated the lowest pro-oxidative marker levels. NPH insulin treatment improved cardiac function, and combination therapy effectively restored cardiac function in diabetic animals.
Conclusions: NPH insulin treatment reduced hyperglycemia and improved cardiac function in diabetic rats. The combined approach of NPH insulin and HBOT resulted in decreased pro-oxidative markers. These findings provide valuable insights for managing cardiovascular complications and oxidative stress in diabetes.
Keywords: hyperbaric oxygen therapy; neutral protamine hagedorn (NPH) insulin; streptozotocin; type 1 diabetes (T1D).
Conflict of interest statement
The authors declare no conflict of interest. The funder had no role in the design of the study, in the collection, analyses, or interpretation of data, in the writing of the manuscript, or in the decision to publish the results.
Figures
Similar articles
-
Hyperbaric Oxygen Therapy Dampens Inflammatory Cytokine Production and Does Not Worsen the Cardiac Function and Oxidative State of Diabetic Rats.Antioxidants (Basel). 2019 Nov 30;8(12):607. doi: 10.3390/antiox8120607. Antioxidants (Basel). 2019. PMID: 31801203 Free PMC article.
-
Effect of chronic administration of PDE5 combined with glycemic control on erectile function in streptozotocin-induced diabetic rats.J Sex Med. 2015 Mar;12(3):600-10. doi: 10.1111/jsm.12752. Epub 2014 Nov 20. J Sex Med. 2015. PMID: 25411013
-
Effect of Hyperbaric Oxygen Therapy (HBOT) on Insulin Resistance Associated with Abdominal Obesity in Wistar Rats with Dietary Sucrose-Induced Metabolic Syndrome.J Nutr Sci Vitaminol (Tokyo). 2021;67(5):292-300. doi: 10.3177/jnsv.67.292. J Nutr Sci Vitaminol (Tokyo). 2021. PMID: 34719614
-
Proliferative retinopathy during hyperbaric oxygen treatment.Diving Hyperb Med. 2017 Sep;47(3):203. doi: 10.28920/dhm47.3.203. Diving Hyperb Med. 2017. PMID: 28868603 Free PMC article. Review.
-
(Ultra-)long-acting insulin analogues for people with type 1 diabetes mellitus.Cochrane Database Syst Rev. 2021 Mar 4;3(3):CD013498. doi: 10.1002/14651858.CD013498.pub2. Cochrane Database Syst Rev. 2021. PMID: 33662147 Free PMC article.
Cited by
-
Effect of GLP-1 Receptor Agonist on Ischemia Reperfusion Injury in Rats with Metabolic Syndrome.Pharmaceuticals (Basel). 2024 Apr 19;17(4):525. doi: 10.3390/ph17040525. Pharmaceuticals (Basel). 2024. PMID: 38675485 Free PMC article.
-
Endothelial Dysfunction and Cardiovascular Disease: Hyperbaric Oxygen Therapy as an Emerging Therapeutic Modality?J Cardiovasc Dev Dis. 2024 Dec 19;11(12):408. doi: 10.3390/jcdd11120408. J Cardiovasc Dev Dis. 2024. PMID: 39728298 Free PMC article. Review.
-
Cell Culture in a Hyperbaric Chamber: A Research Model to Study the Effects of Hyperbarism (Hyperbaric Pressure) on Bone Cell Culture.Cells. 2025 Aug 19;14(16):1287. doi: 10.3390/cells14161287. Cells. 2025. PMID: 40862766 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Medical
Research Materials

