Preclinical Testing Techniques: Paving the Way for New Oncology Screening Approaches
- PMID: 37760435
- PMCID: PMC10526899
- DOI: 10.3390/cancers15184466
Preclinical Testing Techniques: Paving the Way for New Oncology Screening Approaches
Abstract
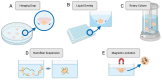
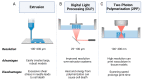
Prior to clinical trials, preclinical testing of oncology drug candidates is performed by evaluating drug candidates with in vitro and in vivo platforms. For in vivo testing, animal models are used to evaluate the toxicity and efficacy of drug candidates. However, animal models often display poor translational results as many drugs that pass preclinical testing fail when tested with humans, with oncology drugs exhibiting especially poor acceptance rates. The FDA Modernization Act 2.0 promotes alternative preclinical testing techniques, presenting the opportunity to use higher complexity in vitro models as an alternative to in vivo testing, including three-dimensional (3D) cell culture models. Three-dimensional tissue cultures address many of the shortcomings of 2D cultures by more closely replicating the tumour microenvironment through a combination of physiologically relevant drug diffusion, paracrine signalling, cellular phenotype, and vascularization that can better mimic native human tissue. This review will discuss the common forms of 3D cell culture, including cell spheroids, organoids, organs-on-a-chip, and 3D bioprinted tissues. Their advantages and limitations will be presented, aiming to discuss the use of these 3D models to accurately represent human tissue and as an alternative to animal testing. The use of 3D culture platforms for preclinical drug development is expected to accelerate as these platforms continue to improve in complexity, reliability, and translational predictivity.
Keywords: 3D bioprinting; 3D culture; drug screening; oncology; organ-on-a-chip; organoid; preclinical; spheroid.
Conflict of interest statement
K.V. is the CEO of VoxCell BioInnovation Inc., Victoria, BC, Canada.
Figures

Similar articles
-
Vascularization Strategies in 3D Cell Culture Models: From Scaffold-Free Models to 3D Bioprinting.Int J Mol Sci. 2022 Nov 23;23(23):14582. doi: 10.3390/ijms232314582. Int J Mol Sci. 2022. PMID: 36498908 Free PMC article. Review.
-
A comprehensive review on 3D tissue models: Biofabrication technologies and preclinical applications.Biomaterials. 2024 Jan;304:122408. doi: 10.1016/j.biomaterials.2023.122408. Epub 2023 Nov 27. Biomaterials. 2024. PMID: 38041911 Free PMC article. Review.
-
Could 3D models of cancer enhance drug screening?Biomaterials. 2020 Feb;232:119744. doi: 10.1016/j.biomaterials.2019.119744. Epub 2019 Dec 26. Biomaterials. 2020. PMID: 31918229 Review.
-
3D Engineering of Ocular Tissues for Disease Modeling and Drug Testing.Adv Exp Med Biol. 2019;1186:171-193. doi: 10.1007/978-3-030-28471-8_7. Adv Exp Med Biol. 2019. PMID: 31654390
-
Organ-on-chip models: Implications in drug discovery and clinical applications.J Cell Physiol. 2019 Jun;234(6):8352-8380. doi: 10.1002/jcp.27729. Epub 2018 Nov 15. J Cell Physiol. 2019. PMID: 30443904 Review.
Cited by
-
Ultrasound-Mediated Drug Diffusion, Uptake, and Cytotoxicity in a Glioblastoma 3D Tumour Sphere Model.Cells. 2025 Jun 11;14(12):886. doi: 10.3390/cells14120886. Cells. 2025. PMID: 40558513 Free PMC article.
-
Fluidic Programmable Gravi-maze Array for High Throughput Multiorgan Drug Testing.bioRxiv [Preprint]. 2025 Jun 24:2025.06.18.660241. doi: 10.1101/2025.06.18.660241. bioRxiv. 2025. PMID: 40666863 Free PMC article. Preprint.
-
Molecular docking and biological evaluation of a novel IWS1 inhibitor for the treatment of human retroperitoneal liposarcoma.Sci Rep. 2025 Jul 2;15(1):22965. doi: 10.1038/s41598-025-07215-y. Sci Rep. 2025. PMID: 40594510 Free PMC article.
-
Generation and maintenance of kidney and kidney cancer organoids from patient-derived material for drug development and precision oncology.Mol Ther Methods Clin Dev. 2024 Nov 5;32(4):101368. doi: 10.1016/j.omtm.2024.101368. eCollection 2024 Dec 12. Mol Ther Methods Clin Dev. 2024. PMID: 39659758 Free PMC article.
-
Acoustic Bioprinting: A Glimpse Into an Emerging Field.Small Methods. 2025 Jul 26:e2500733. doi: 10.1002/smtd.202500733. Online ahead of print. Small Methods. 2025. PMID: 40713816 Review.
References
Publication types
LinkOut - more resources
Full Text Sources

