Changes in the Acetylcholinesterase Enzymatic Activity in Tumor Development and Progression
- PMID: 37760598
- PMCID: PMC10526250
- DOI: 10.3390/cancers15184629
Changes in the Acetylcholinesterase Enzymatic Activity in Tumor Development and Progression
Abstract
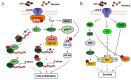
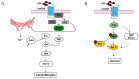
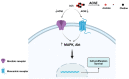
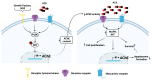
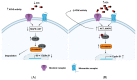
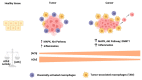
Acetylcholinesterase is a well-known protein because of the relevance of its enzymatic activity in the hydrolysis of acetylcholine in nerve transmission. In addition to the catalytic action, it exerts non-catalytic functions; one is associated with apoptosis, in which acetylcholinesterase could significantly impact the survival and aggressiveness observed in cancer. The participation of AChE as part of the apoptosome could explain the role in tumors, since a lower AChE content would increase cell survival due to poor apoptosome assembly. Likewise, the high Ach content caused by the reduction in enzymatic activity could induce cell survival mediated by the overactivation of acetylcholine receptors (AChR) that activate anti-apoptotic pathways. On the other hand, in tumors in which high enzymatic activity has been observed, AChE could be playing a different role in the aggressiveness of cancer; in this review, we propose that AChE could have a pro-inflammatory role, since the high enzyme content would cause a decrease in ACh, which has also been shown to have anti-inflammatory properties, as discussed in this review. In this review, we analyze the changes that the enzyme could display in different tumors and consider the different levels of regulation that the acetylcholinesterase undergoes in the control of epigenetic changes in the mRNA expression and changes in the enzymatic activity and its molecular forms. We focused on explaining the relationship between acetylcholinesterase expression and its activity in the biology of various tumors. We present up-to-date knowledge regarding this fascinating enzyme that is positioned as a remarkable target for cancer treatment.
Keywords: acetylcholinesterase; apoptosis; cancer; enzymatic activity.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
Regulation of acetylcholinesterase during the lipopolysaccharide-induced inflammatory responses in microglial cells.FASEB J. 2022 Mar;36(3):e22189. doi: 10.1096/fj.202101302RR. FASEB J. 2022. PMID: 35129858
-
Acetylcholinesterase, pro-inflammatory cytokines, and association of ACHE SNP rs 17228602 with male infertility.PLoS One. 2023 Apr 7;18(4):e0282579. doi: 10.1371/journal.pone.0282579. eCollection 2023. PLoS One. 2023. PMID: 37027384 Free PMC article.
-
Possible role of acetylcholinesterase in regulation of postsynaptic receptor efficacy at a central inhibitory synapse of Aplysia.Nature. 1983 Feb 24;301(5902):710-2. doi: 10.1038/301710a0. Nature. 1983. PMID: 6298631
-
Modulators of Acetylcholinesterase Activity: From Alzheimer's Disease to Anti-Cancer Drugs.Curr Med Chem. 2017;24(30):3283-3309. doi: 10.2174/0929867324666170705123509. Curr Med Chem. 2017. PMID: 28685687 Review.
-
Acetylcholinesterase and human cancers.Adv Cancer Res. 2021;152:1-66. doi: 10.1016/bs.acr.2021.05.001. Epub 2021 Jun 21. Adv Cancer Res. 2021. PMID: 34353435 Review.
Cited by
-
Anti-carcinogenic effects of arecaidine but-2-ynyl ester tosylate on breast cancer: proliferation inhibition and activation of apoptosis.Mol Biol Rep. 2025 Mar 4;52(1):278. doi: 10.1007/s11033-025-10385-7. Mol Biol Rep. 2025. PMID: 40035899
-
Acetylcholinesterase enzyme among cancer patients a potential diagnostic and prognostic indicator a multicenter case-control study.Sci Rep. 2024 Mar 1;14(1):5127. doi: 10.1038/s41598-024-55604-6. Sci Rep. 2024. PMID: 38429330 Free PMC article.
-
Diethyl nitrosamine-induces neurobehavioral deficit, oxido-nitrosative stress in rats' brain: a neuroprotective role of diphenyl diselenide.BMC Neurosci. 2024 Dec 25;25(1):77. doi: 10.1186/s12868-024-00922-8. BMC Neurosci. 2024. PMID: 39722026 Free PMC article.
-
5'-Guanidino Xylofuranosyl Nucleosides as Novel Types of 5'-Functionalized Nucleosides with Biological Potential.Pharmaceuticals (Basel). 2025 May 16;18(5):734. doi: 10.3390/ph18050734. Pharmaceuticals (Basel). 2025. PMID: 40430552 Free PMC article.
-
Britanin - a beacon of hope against gastrointestinal tumors?World J Clin Oncol. 2024 Apr 24;15(4):523-530. doi: 10.5306/wjco.v15.i4.523. World J Clin Oncol. 2024. PMID: 38689621 Free PMC article. Review.
References
-
- Pérez-Aguilar B., Vidal C.J., Gomez-Olivares J.L., Gerardo-Ramirez M., Gutiérrez-Ruiz M.C., Gomez-Quiroz L.E. Acetylcholinesterase. In: Choi S., editor. Encyclopedia of Signaling Molecules. Springer International Publishing; Cham, Switzerland: 2018. pp. 91–101.
-
- Cheng K., Samimi R., Xie G., Shant J., Drachenberg C., Wade M., Davis R.J., Nomikos G., Raufman J.-P. Acetylcholine Release by Human Colon Cancer Cells Mediates Autocrine Stimulation of Cell Proliferation. Am. J. Physiol. Gastrointest. Liver Physiol. 2008;295:G591–G597. doi: 10.1152/ajpgi.00055.2008. - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

