Identification of an Antimicrobial Peptide from the Venom of the Trinidad Thick-Tailed Scorpion Tityus trinitatis with Potent Activity against ESKAPE Pathogens and Clostridioides difficile
- PMID: 37760701
- PMCID: PMC10525828
- DOI: 10.3390/antibiotics12091404
Identification of an Antimicrobial Peptide from the Venom of the Trinidad Thick-Tailed Scorpion Tityus trinitatis with Potent Activity against ESKAPE Pathogens and Clostridioides difficile
Abstract
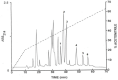
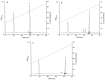
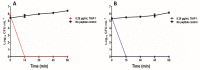
Envenomation by the Trinidad thick-tailed scorpion Tityus trinitatis may result in fatal myocarditis and there is a high incidence of acute pancreatitis among survivors. Peptidomic analysis (reversed-phase HPLC followed by MALDI-TOF mass spectrometry and automated Edman degradation) of T. trinitatis venom led to the isolation and characterization of three peptides with antimicrobial activity. Their primary structures were established asTtAP-1 (FLGSLFSIGSKLLPGVFKLFSRKKQ.NH2), TtAP-2 (IFGMIPGLIGGLISAFK.NH2) and TtAP-3 (FFSLIPSLIGGLVSAIK.NH2). In addition, potassium channel and sodium channel toxins, present in the venom in high abundance, were identified by CID-MS/MS sequence analysis. TtAP-1 was the most potent against a range of clinically relevant Gram-positive and Gram-negative aerobes and against the anaerobe Clostridioides difficile (MIC = 3.1-12.5 µg/mL). At a concentration of 1× MIC, TtAP-1 produced rapid cell death (<15 min against Acinetobacter baumannii and Staphylococcus aureus). The therapeutic potential of TtAP-1 as an anti-infective agent is limited by its high hemolytic activity (LC50 = 18 µg/mL against mouse erythrocytes) but the peptide constitutes a template for the design of analogs that maintain the high bactericidal activity against ESKAPE pathogens but are less toxic to human cells. It is suggested that the antimicrobial peptides in the scorpion venom facilitate the action of the neurotoxins by increasing the membrane permeability of cells from either prey or predator.
Keywords: Clostridioides difficile; ESKAPE pathogen; antimicrobial peptide; neurotoxin; scorpion venom.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study, in the collection, analyses, or interpretation of data, in the writing of the manuscript or in the decision to publish the results.
Figures


Similar articles
-
Meucin-49, a multifunctional scorpion venom peptide with bactericidal synergy with neurotoxins.Amino Acids. 2018 Aug;50(8):1025-1043. doi: 10.1007/s00726-018-2580-0. Epub 2018 May 16. Amino Acids. 2018. PMID: 29770866
-
Repurposing the scorpion venom peptide VmCT1 into an active peptide against Gram-negative ESKAPE pathogens.Bioorg Chem. 2019 Sep;90:103038. doi: 10.1016/j.bioorg.2019.103038. Epub 2019 Jun 8. Bioorg Chem. 2019. PMID: 31212183
-
Multifunctional host-defense peptides isolated from skin secretions of the banana tree dwelling frog Boana platanera (Hylidae; Hylinae).Biochimie. 2024 Aug;223:23-30. doi: 10.1016/j.biochi.2024.03.012. Epub 2024 Mar 30. Biochimie. 2024. PMID: 38561076
-
Biology, venom composition, and scorpionism induced by brazilian scorpion Tityus stigmurus (Thorell, 1876) (Scorpiones: Buthidae): A mini-review.Toxicon. 2020 Oct 15;185:36-45. doi: 10.1016/j.toxicon.2020.06.015. Epub 2020 Jul 6. Toxicon. 2020. PMID: 32585220 Review.
-
Scorpion Venom: Detriments and Benefits.Biomedicines. 2020 May 12;8(5):118. doi: 10.3390/biomedicines8050118. Biomedicines. 2020. PMID: 32408604 Free PMC article. Review.
Cited by
-
A Critical Review of Short Antimicrobial Peptides from Scorpion Venoms, Their Physicochemical Attributes, and Potential for the Development of New Drugs.J Membr Biol. 2024 Aug;257(3-4):165-205. doi: 10.1007/s00232-024-00315-2. Epub 2024 Jul 11. J Membr Biol. 2024. PMID: 38990274 Free PMC article. Review.
-
A New Insight into Phage Combination Therapeutic Approaches Against Drug-Resistant Mixed Bacterial Infections.Phage (New Rochelle). 2024 Dec 18;5(4):203-222. doi: 10.1089/phage.2024.0011. eCollection 2024 Dec. Phage (New Rochelle). 2024. PMID: 40045937 Review.
-
Short Peptides from Asian Scorpions: Bioactive Molecules with Promising Therapeutic Potential.Toxins (Basel). 2025 Feb 28;17(3):114. doi: 10.3390/toxins17030114. Toxins (Basel). 2025. PMID: 40137887 Free PMC article. Review.
-
Scorpion-Induced Acute Coronary Syndrome: A Stinging Complication.J Investig Med High Impact Case Rep. 2024 Jan-Dec;12:23247096241261255. doi: 10.1177/23247096241261255. J Investig Med High Impact Case Rep. 2024. PMID: 38884537 Free PMC article.
References
-
- de Oliveira A.N., Soares A.M., Da Silva S.L. Why to study peptides from venomous and poisonous animals? Int. J. Pept. Res. Ther. 2023;29:76. doi: 10.1007/s10989-023-10543-0. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

