Small Heterodimer Partner Modulates Macrophage Differentiation during Innate Immune Response through the Regulation of Peroxisome Proliferator Activated Receptor Gamma, Mitogen-Activated Protein Kinase, and Nuclear Factor Kappa B Pathways
- PMID: 37760844
- PMCID: PMC10525324
- DOI: 10.3390/biomedicines11092403
Small Heterodimer Partner Modulates Macrophage Differentiation during Innate Immune Response through the Regulation of Peroxisome Proliferator Activated Receptor Gamma, Mitogen-Activated Protein Kinase, and Nuclear Factor Kappa B Pathways
Abstract
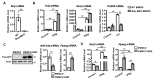
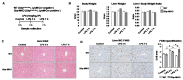
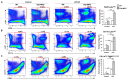
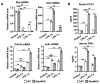
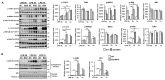
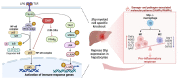
Hepatic macrophages act as the liver's first line of defense against injury. Their differentiation into proinflammatory or anti-inflammatory subpopulations is a critical event that maintains a delicate balance between liver injury and repair. In our investigation, we explored the influence of the small heterodimer partner (SHP), a nuclear receptor primarily associated with metabolism, on macrophage differentiation during the innate immune response. During macrophage differentiation, we observed significant alterations in Shp mRNA expression. Deletion of Shp promoted M1 differentiation while interfering with M2 polarization. Conversely, overexpression of SHP resulted in increased expression of peroxisome proliferator activated receptor gamma (Pparg), a master regulator of anti-inflammatory macrophage differentiation, thereby inhibiting M1 differentiation. Upon lipopolysaccharide (LPS) injection, there was a notable increase in the proinflammatory M1-like macrophages, accompanied by exacerbated infiltration of monocyte-derived macrophages (MDMs) into the livers of Shp myeloid cell specific knockout (Shp-MKO). Concurrently, we observed significant induction of tumor necrosis factor alpha (Tnfa) and chemokine (C-C motif) ligand 2 (Ccl2) expression in LPS-treated Shp-MKO livers. Additionally, the mitogen-activated protein kinase (MAPK) and nuclear factor kappa B (NF-κB) pathways were activated in LPS-treated Shp-MKO livers. Consistently, both pathways were hindered in SHP overexpression macrophages. Finally, we demonstrated that SHP interacts with p65, thereby influencing macrophage immune repones. In summary, our study uncovered a previously unrecognized role of SHP in promoting anti-inflammatory macrophage differentiation during the innate immune response. This was achieved by SHP acting as a regulator for the Pparg, MAPK, and NF-κB pathways.
Keywords: differentiation; knockout; macrophage; nuclear receptor; small heterodimer partner (SHP).
Conflict of interest statement
The authors declare that they have no conflict of interest with the contents of this article.
Figures

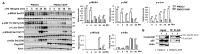
Similar articles
-
Glycogen synthase kinase 3β promotes liver innate immune activation by restraining AMP-activated protein kinase activation.J Hepatol. 2018 Jul;69(1):99-109. doi: 10.1016/j.jhep.2018.01.036. Epub 2018 Feb 13. J Hepatol. 2018. PMID: 29452207 Free PMC article.
-
Hepatocyte nuclear receptor SHP suppresses inflammation and fibrosis in a mouse model of nonalcoholic steatohepatitis.J Biol Chem. 2018 Jun 1;293(22):8656-8671. doi: 10.1074/jbc.RA117.001653. Epub 2018 Apr 17. J Biol Chem. 2018. PMID: 29666185 Free PMC article.
-
Disruption of hepatic small heterodimer partner induces dissociation of steatosis and inflammation in experimental nonalcoholic steatohepatitis.J Biol Chem. 2020 Jan 24;295(4):994-1008. doi: 10.1074/jbc.RA119.010233. Epub 2019 Dec 12. J Biol Chem. 2020. PMID: 31831621 Free PMC article.
-
Antagonistic crosstalk between NF-κB and SIRT1 in the regulation of inflammation and metabolic disorders.Cell Signal. 2013 Oct;25(10):1939-48. doi: 10.1016/j.cellsig.2013.06.007. Epub 2013 Jun 11. Cell Signal. 2013. PMID: 23770291 Review.
-
Postprandial lipoproteins and the molecular regulation of vascular homeostasis.Prog Lipid Res. 2013 Oct;52(4):446-64. doi: 10.1016/j.plipres.2013.06.001. Epub 2013 Jun 15. Prog Lipid Res. 2013. PMID: 23774609 Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

