Omega-3 Fatty Acids Attenuate Renal Fibrosis via AMPK-Mediated Autophagy Flux Activation
- PMID: 37760994
- PMCID: PMC10525956
- DOI: 10.3390/biomedicines11092553
Omega-3 Fatty Acids Attenuate Renal Fibrosis via AMPK-Mediated Autophagy Flux Activation
Abstract
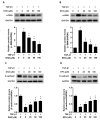
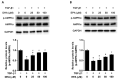
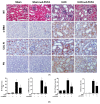
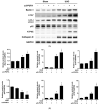
The unilateral ureteral obstruction (UUO) injury model is well-known to mimic human chronic kidney disease, promoting the rapid onset and development of kidney injury. ω3-poly unsaturated fatty acids (PUFAs) have been observed to protect against tissue injury in many disease models. In this study, we assessed the efficacy of ω3-PUFAs in attenuating UUO injury and investigated their mechanism of action. The immortalized human proximal tubular cells human kidney-2 (HK2) were incubated for 72 h with docosahexaenoic acid (DHA) or eicosapentaenoic acid (EPA) in various concentrations, in the presence or absence of transforming growth factor (TGF)-β. DHA/EPA reduced the epithelial-mesenchymal transition in the TGF-β-treated HK2 cells by enhancing autophagy flux and adenosine monophosphate-activated protein kinase (AMPK) phosphorylation. C57BL/6 mice were divided into four groups and treated as follows: sham (no treatment, n = 5), sham + ω3-PUFAs (n = 5), UUO (n = 10), and UUO + ω3-PUFAs (n = 10). Their kidneys and blood were harvested on the seventh day following UUO injury. The kidneys of the ω3-PUFAs-treated UUO mice showed less oxidative stress, inflammation, and fibrosis compared to those of the untreated UUO mice. Greater autophagic flux, higher amounts of microtubule-associated protein 1A/1B-light chain 3 (LC3)-II, Beclin-1, and Atg7, lower amounts of p62, and higher levels of cathepsin D and ATP6E were observed in the kidneys of the omega-3-treated UUO mice compared to those of the control UUO mice. In conclusion, ω3-PUFAs enhanced autophagic activation, leading to a renoprotective response against chronic kidney injury.
Keywords: UUO; autophagy flux; renal fibrosis; ω3-PUFA.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






References
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

