New Formulation of Platelet-Rich Plasma Enriched in Platelet and Extraplatelet Biomolecules Using Hydrogels
- PMID: 37762114
- PMCID: PMC10530784
- DOI: 10.3390/ijms241813811
New Formulation of Platelet-Rich Plasma Enriched in Platelet and Extraplatelet Biomolecules Using Hydrogels
Abstract
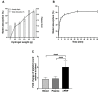
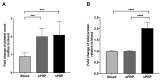
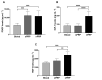
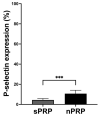
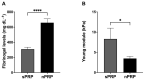
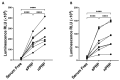
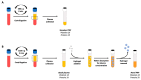
Platelet-rich plasma (PRP) is an autologous biologic product used in several fields of medicine for tissue repair due to the regenerative capacity of the biomolecules of its formulation. PRP consists of a plasma with a platelet concentration higher than basal levels but with basal levels of any biomolecules present out of the platelets. Plasma contains extraplatelet biomolecules known to enhance its regenerative properties. Therefore, a PRP containing not only a higher concentration of platelets but also a higher concentration of extraplatelet biomolecules that could have a stronger regenerative performance than a standard PRP. Considering this, the aim of this work is to develop a new method to obtain PRP enriched in both platelet and extraplatelet molecules. The method is based on the absorption of the water of the plasma using hydroxyethyl acrylamide (HEAA)-based hydrogels. A plasma fraction obtained from blood, containing the basal levels of platelets and proteins, was placed in contact with the HEAA hydrogel powder to absorb half the volume of the water. The resulting plasma was characterized, and its bioactivity was analyzed in vitro. The novel PRP (nPRP) showed a platelet concentration and platelet derived growth factor (PDGF) levels similar to the standard PRP (sPRP), but the concentration of the extraplatelet growth factors IGF-1 (p < 0.0001) and HGF (p < 0.001) were significantly increased. Additionally, the cells exposed to the nPRP showed increased cell viability than those exposed to a sPRP in human dermal fibroblasts (p < 0.001) and primary chondrocytes (p < 0.01). In conclusion, this novel absorption-based method produces a PRP with novel characteristics compared to the standard PRPs, with promising in vitro results that could potentially trigger improved tissue regeneration capacity.
Keywords: biomolecules; growth factors; hydrogel; platelet-rich plasma; platelets; water absorption.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of this study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures

References
-
- Losina E., Paltiel A.D., Weinstein A.M., Yelin E., Hunter D.J., Chen S.P., Klara K., Suter L.G., Solomon D.H., Burbine S.A., et al. Lifetime Medical Costs of Knee Osteoarthritis Management in the United States: Impact of Extending Indications for Total Knee Arthroplasty. Arthritis Care Res. 2015;67:203–215. doi: 10.1002/acr.22412. - DOI - PMC - PubMed
-
- Sánchez M., Delgado D., Garate A., Sánchez P., Oraa J., Bilbao A.M., Guadilla J., Aizpurua B., Fiz N., Azofra J., et al. PRP Injections in Orthopaedic Surgery: Why, When and How to Use PRP Dynamic Liquid Scaffold Injections in Orthopaedic Surgery. IntechOpen; London, UK: 2018.
Grants and funding
- PID2020-120313GB-I00/AIE/10.13039/501100011033/Ministerio de Ciencia y Educación de España
- IT1633-22/Gobierno Vasco Dpto. de Educación
- PIFIND19/03/Ayudas para la formación de personal investigador en colaboración con instituciones y empresas
- PTQ- 3122019-010386/Spanish Ministerio de Economía y Competitividad
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

