Piperine Induces Apoptosis and Autophagy in HSC-3 Human Oral Cancer Cells by Regulating PI3K Signaling Pathway
- PMID: 37762259
- PMCID: PMC10530752
- DOI: 10.3390/ijms241813949
Piperine Induces Apoptosis and Autophagy in HSC-3 Human Oral Cancer Cells by Regulating PI3K Signaling Pathway
Abstract
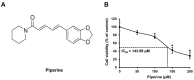
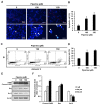
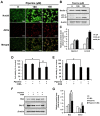
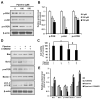
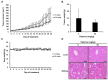
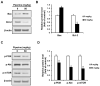
Currently, therapies for treating oral cancer have various side effects; therefore, research on treatment methods employing natural substances is being conducted. This study aimed to investigate piperine-induced apoptosis and autophagy in HSC-3 human oral cancer cells and their effects on tumor growth in vivo. A 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay demonstrated that piperine reduced the viability of HSC-3 cells and 4',6-diamidino-2-phenylindole staining, annexin-V/propidium iodide staining, and analysis of apoptosis-related protein expression confirmed that piperine induces apoptosis in HSC-3 cells. Additionally, piperine-induced autophagy was confirmed by the observation of increased acidic vesicular organelles and autophagy marker proteins, demonstrating that autophagy in HSC-3 cells induces apoptosis. Mechanistically, piperine induced apoptosis and autophagy by inhibiting the phosphatidylinositol-3-kinase (PI3K)/protein kinase B/mammalian target of rapamycin pathway in HSC-3 cells. We also confirmed that piperine inhibits oral cancer tumor growth in vivo via antitumor effects related to apoptosis and PI3K signaling pathway inhibition. Therefore, we suggest that piperine can be considered a natural anticancer agent for human oral cancer.
Keywords: PI3K/Akt/mTOR pathway; anticancer effects; apoptosis; autophagy; oral cancer; piperine.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
The Promise of Piperine in Cancer Chemoprevention.Cancers (Basel). 2023 Nov 20;15(22):5488. doi: 10.3390/cancers15225488. Cancers (Basel). 2023. PMID: 38001748 Free PMC article. Review.
-
Piperlongumine induces apoptosis and autophagy via the PI3K/Akt/mTOR pathway in KB human cervical cancer cells.Food Chem Toxicol. 2023 Oct;180:114051. doi: 10.1016/j.fct.2023.114051. Epub 2023 Sep 19. Food Chem Toxicol. 2023. PMID: 37734464
-
Effects of endoplasmic reticulum stress on the autophagy, apoptosis, and chemotherapy resistance of human breast cancer cells by regulating the PI3K/AKT/mTOR signaling pathway.Tumour Biol. 2017 May;39(5):1010428317697562. doi: 10.1177/1010428317697562. Tumour Biol. 2017. PMID: 28459209
-
Myricetin induces apoptosis and autophagy in human gastric cancer cells through inhibition of the PI3K/Akt/mTOR pathway.Heliyon. 2022 Apr 22;8(5):e09309. doi: 10.1016/j.heliyon.2022.e09309. eCollection 2022 May. Heliyon. 2022. PMID: 35521506 Free PMC article.
-
Arctigenin Triggers Apoptosis and Autophagy via PI3K/Akt/mTOR Inhibition in PC-3M Cells.Chem Pharm Bull (Tokyo). 2021 May 1;69(5):472-480. doi: 10.1248/cpb.c21-00021. Epub 2021 Feb 25. Chem Pharm Bull (Tokyo). 2021. PMID: 33627540
Cited by
-
The Promise of Piperine in Cancer Chemoprevention.Cancers (Basel). 2023 Nov 20;15(22):5488. doi: 10.3390/cancers15225488. Cancers (Basel). 2023. PMID: 38001748 Free PMC article. Review.
-
Piperine enhances doxorubicin sensitivity in triple-negative breast cancer by targeting the PI3K/Akt/mTOR pathway and cancer stem cells.Sci Rep. 2024 Aug 6;14(1):18181. doi: 10.1038/s41598-024-65508-0. Sci Rep. 2024. PMID: 39107323 Free PMC article.
-
Cancer chemoprevention: signaling pathways and strategic approaches.Signal Transduct Target Ther. 2025 Apr 18;10(1):113. doi: 10.1038/s41392-025-02167-1. Signal Transduct Target Ther. 2025. PMID: 40246868 Free PMC article. Review.
-
Therapeutic Targeting of Apoptosis, Autophagic Cell Death, Necroptosis, Pyroptosis, and Ferroptosis Pathways in Oral Squamous Cell Carcinoma: Molecular Mechanisms and Potential Strategies.Biomedicines. 2025 Jul 16;13(7):1745. doi: 10.3390/biomedicines13071745. Biomedicines. 2025. PMID: 40722814 Free PMC article. Review.
-
Epigenetic targeting of autophagy for cancer: DNA and RNA methylation.Front Oncol. 2023 Dec 8;13:1290330. doi: 10.3389/fonc.2023.1290330. eCollection 2023. Front Oncol. 2023. PMID: 38148841 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

