Immunogenicity and Cross-Protective Efficacy Induced by an Inactivated Recombinant Avian Influenza A/H5N1 (Clade 2.3.4.4b) Vaccine against Co-Circulating Influenza A/H5Nx Viruses
- PMID: 37766075
- PMCID: PMC10538193
- DOI: 10.3390/vaccines11091397
Immunogenicity and Cross-Protective Efficacy Induced by an Inactivated Recombinant Avian Influenza A/H5N1 (Clade 2.3.4.4b) Vaccine against Co-Circulating Influenza A/H5Nx Viruses
Abstract
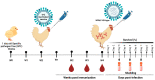
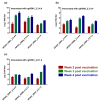
Controlling avian influenza viruses (AIVs) is mainly based on culling of the infected bird flocks or via the implementation of inactivated vaccines in countries where AIVs are considered to be endemic. Over the last decade, several avian influenza virus subtypes, including highly pathogenic avian influenza (HPAI) H5N1 clade 2.2.1.2, H5N8 clade 2.3.4.4b and the recent H5N1 clade 2.3.4.4b, have been reported among poultry populations in Egypt. This demanded the utilization of a nationwide routine vaccination program in the poultry sector. Antigenic differences between available avian influenza vaccines and the currently circulating H5Nx strains were reported, calling for an updated vaccine for homogenous strains. In this study, three H5Nx vaccines were generated by utilizing the reverse genetic system: rgH5N1_2.3.4.4, rgH5N8_2.3.4.4 and rgH5N1_2.2.1.2. Further, the immunogenicity and the cross-reactivity of the generated inactivated vaccines were assessed in the chicken model against a panel of homologous and heterologous H5Nx HPAIVs. Interestingly, the rgH5N1_2.3.4.4 induced high immunogenicity in specific-pathogen-free (SPF) chicken and could efficiently protect immunized chickens against challenge infection with HPAIV H5N1_2.3.4.4, H5N8_2.3.4.4 and H5N1_2.2.1.2. In parallel, the rgH5N1_2.2.1.2 could partially protect SPF chickens against infection with HPAIV H5N1_2.3.4.4 and H5N8_2.3.4.4. Conversely, the raised antibodies to rgH5N1_2.3.4.4 could provide full protection against HPAIV H5N1_2.3.4.4 and HPAIV H5N8_2.3.4.4, and partial protection (60%) against HPAIV H5N1_2.2.1.2. Compared to rgH5N8_2.3.4.4 and rgH5N1_2.2.1.2 vaccines, chickens vaccinated with rgH5N1_2.3.4.4 showed lower viral shedding following challenge infection with the predefined HPAIVs. These data emphasize the superior immunogenicity and cross-protective efficacy of the rgH5N1_2.3.4.4 in comparison to rgH5N8_2.3.4.4 and rgH5N1_2.2.1.2.
Keywords: AIV H5N1; AIV H5N8; clade 2.3.4.4b; highly pathogenic avian influenza; immunogenicity; vaccine.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures

Similar articles
-
Examination of the protective efficacy of two avian influenza H5 vaccines against clade 2.3.4.4b H5N8 highly pathogenic avian influenza virus in commercial broilers.Res Vet Sci. 2021 Nov;140:125-133. doi: 10.1016/j.rvsc.2021.08.012. Epub 2021 Aug 17. Res Vet Sci. 2021. PMID: 34425414
-
The efficacy of an inactivated avian influenza H5N1 vaccine against an African strain of HPAI H5N8 (clade 2.3.4.4 B).Avian Pathol. 2023 Jun;52(3):176-184. doi: 10.1080/03079457.2023.2181145. Epub 2023 May 2. Avian Pathol. 2023. PMID: 37079321
-
Multivalent Inactivated Vaccine Protects Chickens from Distinct Clades of Highly Pathogenic Avian Influenza Subtypes H5N1 and H5N8.Vaccines (Basel). 2025 Feb 19;13(2):204. doi: 10.3390/vaccines13020204. Vaccines (Basel). 2025. PMID: 40006750 Free PMC article.
-
The genetics of highly pathogenic avian influenza viruses of subtype H5 in Germany, 2006-2020.Transbound Emerg Dis. 2021 May;68(3):1136-1150. doi: 10.1111/tbed.13843. Epub 2020 Sep 29. Transbound Emerg Dis. 2021. PMID: 32964686 Review.
-
A review of H5Nx avian influenza viruses.Ther Adv Vaccines Immunother. 2019 Feb 22;7:2515135518821625. doi: 10.1177/2515135518821625. eCollection 2019. Ther Adv Vaccines Immunother. 2019. PMID: 30834359 Free PMC article. Review.
Cited by
-
Evaluation of safety, immunogenicity, and efficacy of inactivated reverse-genetics-based H5N8 highly pathogenic avian influenza virus vaccine with various adjuvants via parenteral and mucosal routes in chickens.Front Immunol. 2025 Mar 20;16:1539492. doi: 10.3389/fimmu.2025.1539492. eCollection 2025. Front Immunol. 2025. PMID: 40181968 Free PMC article.
-
Clade 2.3.4.4b but not historical clade 1 HA replicating RNA vaccine protects against bovine H5N1 challenge in mice.Nat Commun. 2025 Jan 14;16(1):655. doi: 10.1038/s41467-024-55546-7. Nat Commun. 2025. PMID: 39809744 Free PMC article.
-
Clade 2.3.4.4b highly pathogenic avian influenza H5N1 viruses: knowns, unknowns, and challenges.J Virol. 2025 Jun 17;99(6):e0042425. doi: 10.1128/jvi.00424-25. Epub 2025 May 9. J Virol. 2025. PMID: 40340397 Free PMC article. Review.
-
Vaccine optimization for highly pathogenic avian influenza: Assessment of antibody responses and protection for virus-like particle vaccines in chickens.Vaccine X. 2024 Sep 6;20:100552. doi: 10.1016/j.jvacx.2024.100552. eCollection 2024 Oct. Vaccine X. 2024. PMID: 39309609 Free PMC article.
-
Immunogenic and Protective Properties of Recombinant Hemagglutinin of Influenza A (H5N8) Virus.Vaccines (Basel). 2024 Jan 29;12(2):143. doi: 10.3390/vaccines12020143. Vaccines (Basel). 2024. PMID: 38400127 Free PMC article.
References
-
- Youk S., Torchetti M.K., Lantz K., Lenoch J.B., Killian M.L., Leyson C., Bevins S.N., Dilione K., Ip H.S., Stallknecht D.E., et al. H5N1 highly pathogenic avian influenza clade 2.3.4.4b in wild and domestic birds: Introductions into the United States and reassortments, December 2021–April 2022. Virology. 2023;587:109860. doi: 10.1016/j.virol.2023.109860. - DOI - PubMed
-
- Mosaad Z., Elhusseiny M.H., Zanaty A., Fathy M.M., Hagag N.M., Mady W.H., Said D., Elsayed M.M., Erfan A.M., Rabie N., et al. Emergence of Highly Pathogenic Avian Influenza A Virus (H5N1) of Clade 2.3.4.4b in Egypt, 2021–2022. Pathogens. 2023;12:90. doi: 10.3390/pathogens12010090. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

