Structural Characterization of Canine Minute Virus, Rat and Porcine Bocavirus
- PMID: 37766206
- PMCID: PMC10534443
- DOI: 10.3390/v15091799
Structural Characterization of Canine Minute Virus, Rat and Porcine Bocavirus
Abstract
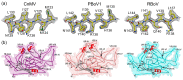
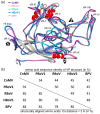
Bocaparvovirus is an expansive genus of the Parvovirinae, with a wide range of vertebrate hosts. This study investigates Canine minute virus (CnMV), Rat bocavirus (RBoV), and Porcine bocavirus 1 (PBoV1). Both CnMV and PBoV1 have been found in gastrointestinal infections in their respective hosts, with CnMV responsible for spontaneous abortions in dogs, while PBoV has been associated with encephalomyelitis in piglets. The pathogenicity of the recently identified RBoV is currently unknown. To initiate the characterization of these viruses, their capsids structures were determined by cryo-electron microscopy at resolutions ranging from 2.3 to 2.7 Å. Compared to other parvoviruses, the CnMV, PBoV1, and RBoV capsids showed conserved features, such as the channel at the fivefold symmetry axis. However, major differences were observed at the two- and threefold axes. While CnMV displays prominent threefold protrusions, the same region is more recessed in PBoV1 and RBoV. Furthermore, the typical twofold axis depression of parvoviral capsids is absent in CnMV or very small in PBoV and RBoV. These capsid structures extend the structural portfolio for the Bocaparvovirus genus and will allow future characterization of these pathogens on a molecular level. This is important, as no antivirals or vaccines exist for these viruses.
Keywords: CnMV; PBoV; RBoV; bocavirus; capsid; cryo-EM; parvovirus; pathogen.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Structural Insights into Human Bocaparvoviruses.J Virol. 2017 May 12;91(11):e00261-17. doi: 10.1128/JVI.00261-17. Print 2017 Jun 1. J Virol. 2017. PMID: 28331084 Free PMC article.
-
Structure of an enteric pathogen, bovine parvovirus.J Virol. 2015 Mar;89(5):2603-14. doi: 10.1128/JVI.03157-14. Epub 2014 Dec 17. J Virol. 2015. PMID: 25520501 Free PMC article.
-
Identification and genomic characterization of a novel rat bocavirus from brown rats in China.Infect Genet Evol. 2017 Jan;47:68-76. doi: 10.1016/j.meegid.2016.11.014. Epub 2016 Nov 19. Infect Genet Evol. 2017. PMID: 27871815
-
Animal bocaviruses: a brief review.Intervirology. 2008;51(5):328-34. doi: 10.1159/000173734. Epub 2008 Nov 20. Intervirology. 2008. PMID: 19023216 Review.
-
Porcine Bocavirus: A 10-Year History since Its Discovery.Virol Sin. 2021 Dec;36(6):1261-1272. doi: 10.1007/s12250-021-00365-z. Epub 2021 Apr 28. Virol Sin. 2021. PMID: 33909219 Free PMC article. Review.
Cited by
-
Birds of a feather flock together: structural characterization of red-crowned crane and turkey aveparvoviruses.J Virol. 2025 Jul 22;99(7):e0011025. doi: 10.1128/jvi.00110-25. Epub 2025 Jul 3. J Virol. 2025. PMID: 40607800 Free PMC article.
References
-
- Penzes J.J., Soderlund-Venermo M., Canuti M., Eis-Hubinger A.M., Hughes J., Cotmore S.F., Harrach B. Reorganizing the family Parvoviridae: A revised taxonomy independent of the canonical approach based on host association. Arch. Virol. 2020;165:2133–2146. doi: 10.1007/s00705-020-04632-4. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

