Non-Polio Enterovirus C Replicate in Both Airway and Intestine Organotypic Cultures
- PMID: 37766230
- PMCID: PMC10537321
- DOI: 10.3390/v15091823
Non-Polio Enterovirus C Replicate in Both Airway and Intestine Organotypic Cultures
Abstract
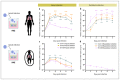
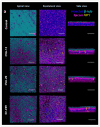
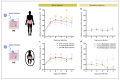
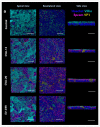
Non-polio enteroviruses (EV) belonging to species C, which are highly prevalent in Africa, mainly among children, are poorly characterized, and their pathogenesis is mostly unknown as they are difficult to culture. In this study, human airway and intestinal organotypic models were used to investigate tissue and cellular tropism of three EV-C genotypes, EV-C99, CVA-13, and CVA-20. Clinical isolates were obtained within the two passages of culture on Caco2 cells, and all three viruses were replicated in both the human airway and intestinal organotypic cultures. We did not observe differences in viral replication between fetal and adult tissue that could potentially explain the preferential infection of infants by EV-C genotypes. Infection of the airway and the intestinal cultures indicates that they both can serve as entry sites for non-polio EV-C. Ciliated airway cells and enterocytes are the target of infection for all three viruses, as well as enteroendocrine cells for EV-C99.
Keywords: CVA-13; CVA-20; EV-C99; enterovirus C species; human airway epithelium; human intestinal epithelium; non-polio enterovirus; organoids; organotypic cultures; tropism.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






References
-
- [(accessed on 6 February 2023)]. Available online: https://www.picornaviridae.com/
-
- Sawyer M.H. Enterovirus infections: Diagnosis and treatment. Semin. Pediatr. Infect. Dis. 2002;13:40–47. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

