Exosomal non-coding RNAs: Blueprint in colorectal cancer metastasis and therapeutic targets
- PMID: 37767111
- PMCID: PMC10520679
- DOI: 10.1016/j.ncrna.2023.09.001
Exosomal non-coding RNAs: Blueprint in colorectal cancer metastasis and therapeutic targets
Abstract
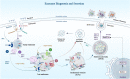
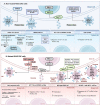
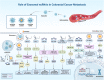
Colorectal cancer (CRC) is ranked as the world's third-most prevalent cancer, and metastatic CRC considerably increases cancer-related fatalities globally. A number of complex mechanisms that are strictly controlled at the molecular level are involved in metastasis, which is the primary reason for death in people with CRC. Recently, it has become clear that exosomes, which are small extracellular vesicles released by non-tumorous and tumorigenic cells, play a critical role as communication mediators among tumor microenvironment (TME). To facilitate communication between the TME and cancer cells, non-coding RNAs (ncRNAs) play a crucial role and are recognized as potent regulators of gene expression and cellular processes, such as metastasis and drug resistance. NcRNAs are now recognized as potent regulators of gene expression and many hallmarks of cancer, including metastasis. Exosomal ncRNAs, like miRNAs, circRNAs, and lncRNAs, have been demonstrated to influence a number of cellular mechanisms that contribute to CRC metastasis. However, the molecular mechanisms that link exosomal ncRNAs with CRC metastasis are not well understood. This review highlights the essential roles that exosomal ncRNAs play in the progression of CRC metastatic disease and explores the therapeutic choices that are open to patients who have CRC metastases. However, exosomal ncRNA treatment strategy development is still in its early phases; consequently, additional investigation is required to improve delivery methods and find novel therapeutic targets as well as confirm the effectiveness and safety of these therapies in preclinical and clinical contexts.
Keywords: Exosomal ncRNAs; Metastatic CRC; Therapeutic targets.
© 2023 The Authors.
Figures

Similar articles
-
The significance of exosomal non-coding RNAs (ncRNAs) in the metastasis of colorectal cancer and development of therapy resistance.Gene. 2025 Feb 10;937:149141. doi: 10.1016/j.gene.2024.149141. Epub 2024 Dec 4. Gene. 2025. PMID: 39643147 Review.
-
Role of Exosomal Non-Coding RNAs In Vivo and In Vitro Studies in Colorectal Cancer.Microrna. 2025 Mar 18. doi: 10.2174/0122115366331268250303031240. Online ahead of print. Microrna. 2025. PMID: 40108898
-
Exosomal non-coding RNAs in colorectal cancer metastasis.Clin Chim Acta. 2024 Mar 15;556:117849. doi: 10.1016/j.cca.2024.117849. Epub 2024 Feb 28. Clin Chim Acta. 2024. PMID: 38417779 Review.
-
The Potential Roles of Exosomal Non-Coding RNAs in Hepatocellular Carcinoma.Front Oncol. 2022 Feb 24;12:790916. doi: 10.3389/fonc.2022.790916. eCollection 2022. Front Oncol. 2022. PMID: 35280805 Free PMC article. Review.
-
Exosomal Non-coding RNAs-Mediated Crosstalk in the Tumor Microenvironment.Front Cell Dev Biol. 2021 Apr 12;9:646864. doi: 10.3389/fcell.2021.646864. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 33912560 Free PMC article. Review.
Cited by
-
Stage-Specific Plasma Metabolomic Profiles in Colorectal Cancer.J Clin Med. 2024 Sep 2;13(17):5202. doi: 10.3390/jcm13175202. J Clin Med. 2024. PMID: 39274416 Free PMC article.
-
The homologous tumor-derived-exosomes loaded with miR-1270 selectively enhanced the suppression effect for colorectal cancer cells.Cancer Med. 2024 Jan;13(1):e6936. doi: 10.1002/cam4.6936. Epub 2024 Jan 10. Cancer Med. 2024. PMID: 38197582 Free PMC article.
-
Exosomal long non-coding RNAs in cancer: Interplay, modulation, and therapeutic avenues.Noncoding RNA Res. 2024 Apr 4;9(3):887-900. doi: 10.1016/j.ncrna.2024.03.014. eCollection 2024 Sep. Noncoding RNA Res. 2024. PMID: 38616862 Free PMC article. Review.
-
Extracellular vesicles isolated from SphK1 inhibitor SKI II-medium restrain the migration of colorectal cancer.Transl Cancer Res. 2025 May 30;14(5):2594-2602. doi: 10.21037/tcr-24-2152. Epub 2025 May 7. Transl Cancer Res. 2025. PMID: 40530117 Free PMC article.
-
Role of exosomal non‑coding RNAs in cancer‑associated fibroblast‑mediated therapy resistance (Review).Int J Oncol. 2025 Aug;67(2):68. doi: 10.3892/ijo.2025.5774. Epub 2025 Jul 19. Int J Oncol. 2025. PMID: 40682844 Free PMC article. Review.
References
-
- Sulekha Suresh D., Guruvayoorappan C. Molecular principles of tissue invasion and metastasis. Am. J. Physiol. Cell Physiol. 2023;324(5):C971–C991. - PubMed
-
- van Niel G., D'Angelo G., Raposo G. Shedding light on the cell biology of extracellular vesicles. Nat. Rev. Mol. Cell Biol. 2018;19(4):213–228. - PubMed
Publication types
LinkOut - more resources
Full Text Sources

