Increasing glutathione levels by a novel posttranslational mechanism inhibits neuronal hyperexcitability
- PMID: 37769522
- PMCID: PMC10539966
- DOI: 10.1016/j.redox.2023.102895
Increasing glutathione levels by a novel posttranslational mechanism inhibits neuronal hyperexcitability
Abstract
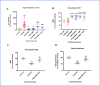
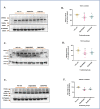
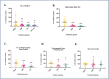
Glutathione (GSH) depletion, and impaired redox homeostasis have been observed in experimental animal models and patients with epilepsy. Pleiotropic strategies that elevate GSH levels via transcriptional regulation have been shown to significantly decrease oxidative stress and seizure frequency, increase seizure threshold, and rescue certain cognitive deficits. Whether elevation of GSH per se alters neuronal hyperexcitability remains unanswered. We previously showed that thiols such as dimercaprol (DMP) elevate GSH via post-translational activation of glutamate cysteine ligase (GCL), the rate limiting GSH biosynthetic enzyme. Here, we asked if elevation of cellular GSH by DMP altered neuronal hyperexcitability in-vitro and in-vivo. Treatment of primary neuronal-glial cerebrocortical cultures with DMP elevated GSH and inhibited a voltage-gated potassium channel blocker (4-aminopyridine, 4AP) induced neuronal hyperexcitability. DMP increased GSH in wildtype (WT) zebrafish larvae and significantly attenuated convulsant pentylenetetrazol (PTZ)-induced acute 'seizure-like' swim behavior. DMP treatment increased GSH and inhibited convulsive, spontaneous 'seizure-like' swim behavior in the Dravet Syndrome (DS) zebrafish larvae (scn1Lab). Furthermore, DMP treatment significantly decreased spontaneous electrographic seizures and associated seizure parameters in scn1Lab zebrafish larvae. We investigated the role of the redox-sensitive mammalian target of rapamycin (mTOR) pathway due to the presence of several cysteine-rich proteins and their involvement in regulating neuronal excitability. Treatment of primary neuronal-glial cerebrocortical cultures with 4AP or l-buthionine-(S,R)-sulfoximine (BSO), an irreversible inhibitor of GSH biosynthesis, significantly increased mTOR complex I (mTORC1) activity which was rescued by pre-treatment with DMP. Furthermore, BSO-mediated GSH depletion oxidatively modified the tuberous sclerosis protein complex (TSC) consisting of hamartin (TSC1), tuberin (TSC2), and TBC1 domain family member 7 (TBC1D7) which are critical negative regulators of mTORC1. In summary, our results suggest that DMP-mediated GSH elevation by a novel post-translational mechanism can inhibit neuronal hyperexcitability both in-vitro and in-vivo and a plausible link is the redox sensitive mTORC1 pathway.
Keywords: Glutamate cysteine ligase; Glutathione; Neuronal hyperexcitability; Oxidative stress; mTORC1.
Copyright © 2023 The Authors. Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures






References
-
- Kwan P., Arzimanoglou A., Berg A.T., et al. Definition of drug resistant epilepsy: consensus proposal by the ad hoc Task Force of the ILAE Commission on Therapeutic Strategies. Epilepsia. 2010;51(6):1069–1077. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

