Cobalt Stabilization through Mesopore Confinement on TiO2 Support for Fischer-Tropsch Reaction
- PMID: 37771503
- PMCID: PMC10523356
- DOI: 10.1021/acsaem.3c01432
Cobalt Stabilization through Mesopore Confinement on TiO2 Support for Fischer-Tropsch Reaction
Abstract
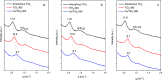
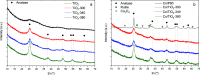
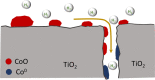
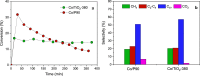
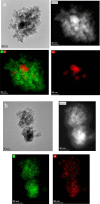
Cobalt supported on mesostructured TiO2 catalysts has been prepared by a wet-impregnation method. The Co/TiO2 catalytic system showed better catalytic performance after support calcination at 380 °C. Co nanoparticles appeared well distributed along the mesopore channels of TiO2. After reduction pretreatment and reaction, a drastic structural change leads to mesopore structure collapse and the dispersion of the Co nanoparticles on the external surface. Along this complex process, Co species first form discrete nanoparticles inside the pore and then diffuse out as the pore collapses. Through this confinement, a strong metal-support interaction effect is hindered, and highly stable metal active sites lead to better performance for Fischer-Tropsch synthesis reaction toward C5+ products.
© 2023 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures








References
-
- Ang T. Z.; Salem M.; Kamarol M.; Das H. S.; Nazari M. A.; Prabaharan N. A comprehensive study of renewable energy sources: Classifications, challenges and suggestions. Energy Strategy Rev. 2022, 43, 100939. 10.1016/j.esr.2022.100939. - DOI
-
- Grim R. G.; To A. T.; Farberow C. A.; Hensley J. E.; Ruddy D. A.; Schaidle J. A. Growing the Bioeconomy through Catalysis: A review of recent advancements in the production of fuels and chemicals from syngas-derived oxygenates. ACS Catal. 2019, 9, 4145–4172. 10.1021/acscatal.8b03945. - DOI
-
- Martinelli M.; Gnanamani M. K.; LeViness S.; Jacobs G.; Shafer W. D. An overview of Fischer–Tropsch Synthesis: XtL processes, catalysts and reactors. Appl. Catal. A: Gen.. 2020, 608, 117740. 10.1016/j.apcata.2020.117740. - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous
