The γ-Adducin 1-357 fragment promotes tau pathology
- PMID: 37771520
- PMCID: PMC10526357
- DOI: 10.3389/fnagi.2023.1241750
The γ-Adducin 1-357 fragment promotes tau pathology
Abstract
Background: Tau phosphorylation is a pathological hallmark of Alzheimer's disease (AD). Previously, we reported that the γ-adducin 1-357 fragment is present in the brains of AD patients. However, it remains unknown how γ-adducin regulates tau phosphorylation.
Objective: The aim of this project is to investigate the effects of the γ-adducin 1-357 fragment on tau phosphorylation and the kinases involved in this process.
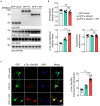
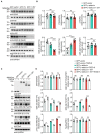
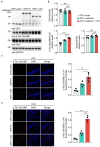
Methods: Full-length γ-adducin or the γ-adducin 1-357 fragment was expressed in HEK293 cells, SH-SY5Y cells, and primary neurons. The phosphorylation of tau Ser396 was determined using Western blot and immunofluorescence. Tau P301S transgenic mice were injected with adeno-associated virus encoding full-length γ-adducin or γ-adducin 1-357 fragment to determine the phosphorylation of tau.
Results: The γ-adducin 1-357 fragment enhances tau phosphorylation at Ser396. Additionally, the expression of the γ-adducin 1-357 fragment leads to the activation of glycogen synthase kinase-3β (GSK-3β). This effect was mitigated by the GSK-3β inhibitor 4-Benzyl-2-methyl-1,2,4-thiadiazolidine-3,5-dione (TDZD-8).
Conclusion: The γ-adducin 1-357 fragment enhances tau phosphorylation by activating GSK3β. These results support that the fragmentation of γ-adducin may play a pivotal role in tau pathology.
Keywords: 4-Benzyl-2-methyl-1,2,4-thiadiazolidine-3,5-dione; Alzheimer’s disease; glycogen synthase kinase-3β; tau phosphorylation; γ-adducin.
Copyright © 2023 Yu, Xiong, Liu, Xia, Meng and Zhang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
TPPU protects tau from H2O2-induced hyperphosphorylation in HEK293/tau cells by regulating PI3K/AKT/GSK-3β pathway.J Huazhong Univ Sci Technolog Med Sci. 2016 Dec;36(6):785-790. doi: 10.1007/s11596-016-1662-z. Epub 2016 Dec 7. J Huazhong Univ Sci Technolog Med Sci. 2016. PMID: 27924507
-
TDZD-8 alleviates delayed neurological sequelae following acute carbon monoxide poisoning involving tau protein phosphorylation.Inhal Toxicol. 2020 Feb;32(2):79-85. doi: 10.1080/08958378.2020.1741739. Epub 2020 Mar 18. Inhal Toxicol. 2020. PMID: 32188325
-
Activation of tau protein kinase I/glycogen synthase kinase-3beta by amyloid beta peptide (25-35) enhances phosphorylation of tau in hippocampal neurons.Neurosci Res. 1998 Aug;31(4):317-23. doi: 10.1016/s0168-0102(98)00061-3. Neurosci Res. 1998. PMID: 9809590
-
Leucine-rich repeat kinase 2 regulates tau phosphorylation through direct activation of glycogen synthase kinase-3β.FEBS J. 2014 Jan;281(1):3-13. doi: 10.1111/febs.12579. Epub 2013 Nov 28. FEBS J. 2014. PMID: 24165324
-
Diabetes mellitus and Alzheimer's disease: GSK-3β as a potential link.Behav Brain Res. 2018 Feb 26;339:57-65. doi: 10.1016/j.bbr.2017.11.015. Epub 2017 Nov 21. Behav Brain Res. 2018. PMID: 29158110 Review.
References
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

