High dose, dual-release polymeric films for extended surgical bed paclitaxel delivery
- PMID: 37776906
- PMCID: PMC10990290
- DOI: 10.1016/j.jconrel.2023.09.048
High dose, dual-release polymeric films for extended surgical bed paclitaxel delivery
Abstract
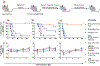
While surgery represents a major therapy for most solid organ cancers, local recurrence is clinically problematic for cancers such as sarcoma for which adjuvant radiotherapy and systemic chemotherapy provide minimal local control or survival benefit and are dose-limited due to off-target side effects. We describe an implantable, biodegradable poly(1,2-glycerol carbonate) and poly(caprolactone) film with entrapped and covalently-bound paclitaxel enabling safe, controlled, and extended local delivery of paclitaxel achieving concentrations 10,000× tissue levels compared to systemic administration. Films containing entrapped and covalently-bound paclitaxel implanted in the tumor bed, immediately after resection of human cell line-derived chondrosarcoma and patient-derived xenograft liposarcoma and leiomyosarcoma in mice, improve median 90- or 200-day recurrence-free and overall survival compared to control mice. Furthermore, mice in the experimental film arm show no film-related morbidity. Continuous, extended, high-dose paclitaxel delivery via this unique polymer platform safely improves outcomes in three different sarcoma models and provides a rationale for future incorporation into human trials.
Keywords: Drug delivery; Dual release; Paclitaxel; Resection; Sarcoma.
Copyright © 2023 Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest CB, YLC, and MWG are co-inventors on a patent application, which is available for licensing. All other authors declare they have no competing interests.
Figures

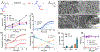
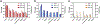
Similar articles
-
Paclitaxel-eluting polymer film reduces locoregional recurrence and improves survival in a recurrent sarcoma model: a novel investigational therapy.Ann Surg Oncol. 2012 Jan;19(1):199-206. doi: 10.1245/s10434-011-1871-4. Epub 2011 Jul 16. Ann Surg Oncol. 2012. PMID: 21769471
-
Prevention of local tumor recurrence following surgery using low-dose chemotherapeutic polymer films.Ann Surg Oncol. 2010 Apr;17(4):1203-13. doi: 10.1245/s10434-009-0856-z. Epub 2009 Dec 3. Ann Surg Oncol. 2010. PMID: 19957041
-
Sustained Supratherapeutic Paclitaxel Delivery Enhances Irreversible Sarcoma Cell Death.Mol Cancer Ther. 2022 Nov 3;21(11):1663-1673. doi: 10.1158/1535-7163.MCT-21-0750. Mol Cancer Ther. 2022. PMID: 36031342 Free PMC article.
-
Innovative strategies for effective paclitaxel delivery: Recent developments and prospects.J Oncol Pharm Pract. 2024 Mar;30(2):367-384. doi: 10.1177/10781552231208978. Epub 2024 Jan 10. J Oncol Pharm Pract. 2024. PMID: 38204196 Review.
-
The paclitaxel (TAXUS)-eluting stent: a review of its use in the management of de novo coronary artery lesions.Am J Cardiovasc Drugs. 2004;4(4):257-68. doi: 10.2165/00129784-200404040-00006. Am J Cardiovasc Drugs. 2004. PMID: 15285700 Review.
References
-
- Siegel RL, Miller KD, Fuchs HE, Jemal A, Cancer statistics, 2022, CA Cancer J Clin. 72 (2022) 7–33. https://doi.org/10.3322/caac.21708. - DOI - PubMed
-
- Stiller CA, Trama A, Serraino D, Rossi S, Navarro C, Chirlaque MD, Casali PG, Descriptive epidemiology of sarcomas in Europe: Report from the RARECARE project, Eur J Cancer. 49 (2013) 684–695. https://doi.org/10.1016/j.ejca.2012.09.011. - DOI - PubMed
-
- Surveillance, Epidemiology, and End Results (SEER) Program (www.seer.cancer.gov) SEER*Stat Database: Mortality - All COD, Aggregated With State, Total U.S. (1969–2019) <Katrina/Rita Population Adjustment>, National Cancer Institute, DCCPS, Surveillance Research Program, released April 2021. Underlying mortality data provided by NCHS (www.cdc.gov/nchs), (n.d.).
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

