Neuronal plasticity contributes to postictal death
- PMID: 37778436
- PMCID: PMC10842614
- DOI: 10.1016/j.pneurobio.2023.102531
Neuronal plasticity contributes to postictal death
Abstract
Repeated generalized tonic-clonic seizures (GTCSs) are the most critical risk factor for sudden unexpected death in epilepsy (SUDEP). GTCSs can cause fatal apnea. We investigated neuronal plasticity mechanisms that precipitate postictal apnea and seizure-induced death. Repeated seizures worsened behavior, precipitated apnea, and enlarged active neuronal circuits, recruiting more neurons in such brainstem nuclei as periaqueductal gray (PAG) and dorsal raphe, indicative of brainstem plasticity. Seizure-activated neurons are more excitable and have enhanced AMPA-mediated excitatory transmission after a seizure. Global deletion of the GluA1 subunit of AMPA receptors abolishes postictal apnea and seizure-induced death. Treatment with a drug that blocks Ca2+-permeable AMPA receptors also renders mice apnea-free with five-fold better survival than untreated mice. Repeated seizures traffic the GluA1 subunit-containing AMPA receptors to synapses, and blocking this mechanism decreases the probability of postictal apnea and seizure-induced death.
Keywords: AMPA; Apnea; Brainstem plasticity; Epilepsy; GluR1 subunit; SUDEP; Seizures.
Copyright © 2023 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures

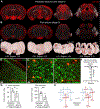


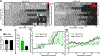
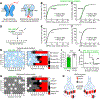
References
-
- Bertram E, 2007. The relevance of kindling for human epilepsy. Epilepsia 48, 65–74. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

