Temporal and spatial properties of vestibular signals for perception of self-motion
- PMID: 37780704
- PMCID: PMC10534010
- DOI: 10.3389/fneur.2023.1266513
Temporal and spatial properties of vestibular signals for perception of self-motion
Abstract
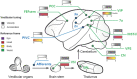
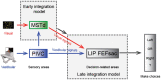
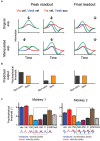
It is well recognized that the vestibular system is involved in numerous important cognitive functions, including self-motion perception, spatial orientation, locomotion, and vector-based navigation, in addition to basic reflexes, such as oculomotor or body postural control. Consistent with this rationale, vestibular signals exist broadly in the brain, including several regions of the cerebral cortex, potentially allowing tight coordination with other sensory systems to improve the accuracy and precision of perception or action during self-motion. Recent neurophysiological studies in animal models based on single-cell resolution indicate that vestibular signals exhibit complex spatiotemporal dynamics, producing challenges in identifying their exact functions and how they are integrated with other modality signals. For example, vestibular and optic flow could provide congruent and incongruent signals regarding spatial tuning functions, reference frames, and temporal dynamics. Comprehensive studies, including behavioral tasks, neural recording across sensory and sensory-motor association areas, and causal link manipulations, have provided some insights into the neural mechanisms underlying multisensory self-motion perception.
Keywords: heading; optic flow; rotation–linear motion; self-motion; vestibular.
Copyright © 2023 Liu, Shan and Gu.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Vestibular System and Self-Motion.Front Cell Neurosci. 2018 Nov 22;12:456. doi: 10.3389/fncel.2018.00456. eCollection 2018. Front Cell Neurosci. 2018. PMID: 30524247 Free PMC article. Review.
-
Vestibular signals in primate cortex for self-motion perception.Curr Opin Neurobiol. 2018 Oct;52:10-17. doi: 10.1016/j.conb.2018.04.004. Epub 2018 Apr 23. Curr Opin Neurobiol. 2018. PMID: 29694922 Review.
-
Heading Tuning in Macaque Area V6.J Neurosci. 2015 Dec 16;35(50):16303-14. doi: 10.1523/JNEUROSCI.2903-15.2015. J Neurosci. 2015. PMID: 26674858 Free PMC article.
-
Spatial reference frames of visual, vestibular, and multimodal heading signals in the dorsal subdivision of the medial superior temporal area.J Neurosci. 2007 Jan 17;27(3):700-12. doi: 10.1523/JNEUROSCI.3553-06.2007. J Neurosci. 2007. PMID: 17234602 Free PMC article.
-
Robust vestibular self-motion signals in macaque posterior cingulate region.Elife. 2021 Apr 8;10:e64569. doi: 10.7554/eLife.64569. Elife. 2021. PMID: 33827753 Free PMC article.
References
-
- Gallistel CR. The organization of learning. Cambridge, MA: The MIT Press; (1990). 648 p.
Publication types
LinkOut - more resources
Full Text Sources

