Temporal Trends in Phenotypic Macrolide and Nonmacrolide Resistance for Streptococcus pneumoniae Nasopharyngeal Samples Up to 36 Months after Mass Azithromycin Administration in a Cluster-Randomized Trial in Niger
- PMID: 37783458
- PMCID: PMC10622462
- DOI: 10.4269/ajtmh.23-0431
Temporal Trends in Phenotypic Macrolide and Nonmacrolide Resistance for Streptococcus pneumoniae Nasopharyngeal Samples Up to 36 Months after Mass Azithromycin Administration in a Cluster-Randomized Trial in Niger
Abstract
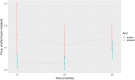
Azithromycin mass drug administration decreases child mortality but also selects for antibiotic resistance. Herein, we evaluate macrolide resistance of nasopharyngeal Streptococcus pneumoniae after azithromycin MDA. In a cluster-randomized trial, children 1-59 months received azithromycin or placebo biannually. Fifteen villages from each arm were randomly selected for antimicrobial resistance testing, and 10-15 randomly selected swabs from enrolled children at each village were processed for S. pneumoniae isolation and resistance testing. The primary prespecified outcome was macrolide resistance fraction for azithromycin versus placebo villages at 36 months. Secondary non-prespecified outcomes were comparisons of azithromycin and placebo for: 1) macrolide resistance at 12, 24, and 36 months; 2) nonmacrolide resistance at 36 months; and 3) suspected-erm mutation. At 36 months, 423 swabs were obtained and 322 grew S. pneumoniae, (azithromycin: 146/202, placebo: 176/221). Mean resistance prevalence was non-significantly higher in treatment than placebo (mixed-effects model: 14.6% vs. 8.9%; OR = 2.0, 95% CI: 0.99-3.97). However, when all time points were evaluated, macrolide resistance prevalence was significantly higher in the azithromycin group (β = 0.102, 95% CI: 0.04-0.167). For all nonmacrolides, resistance prevalence at 36 months was not different between the two groups. Azithromycin and placebo were not different for suspected-erm mutation prevalence. Macrolide resistance was higher in the azithromycin group over all time points, but not at 36 months. Although this suggests resistance may not continue to increase after biannual MDA, more studies are needed to clarify when MDA can safely decrease mortality and morbidity in lower- and middle-income countries.
Figures
Similar articles
-
Long-term effects of azithromycin mass administration to reduce childhood mortality on Streptococcus pneumoniae antimicrobial resistance: a population-based, cross-sectional, follow-up carriage survey.Lancet Infect Dis. 2025 Jun 2:S1473-3099(25)00212-9. doi: 10.1016/S1473-3099(25)00212-9. Online ahead of print. Lancet Infect Dis. 2025. PMID: 40473452
-
Macrolide antibiotics (including azithromycin) for cystic fibrosis.Cochrane Database Syst Rev. 2024 Feb 27;2(2):CD002203. doi: 10.1002/14651858.CD002203.pub5. Cochrane Database Syst Rev. 2024. PMID: 38411248 Free PMC article.
-
Prophylactic antibiotics for adults with chronic obstructive pulmonary disease: a network meta-analysis.Cochrane Database Syst Rev. 2021 Jan 15;1(1):CD013198. doi: 10.1002/14651858.CD013198.pub2. Cochrane Database Syst Rev. 2021. PMID: 33448349 Free PMC article.
-
Macrolide antibiotics for bronchiectasis.Cochrane Database Syst Rev. 2018 Mar 15;3(3):CD012406. doi: 10.1002/14651858.CD012406.pub2. Cochrane Database Syst Rev. 2018. PMID: 29543980 Free PMC article.
-
Spillover of Azithromycin Mass Drug Administration and Child Survival: A Secondary Analysis of a Cluster-Randomized Clinical Trial.JAMA Netw Open. 2025 Jul 1;8(7):e2519693. doi: 10.1001/jamanetworkopen.2025.19693. JAMA Netw Open. 2025. PMID: 40638118 Free PMC article. Clinical Trial.
Cited by
-
Mass drug administration trials of azithromycin: an analysis to inform future research and guidelines.Infect Dis Poverty. 2025 Jul 21;14(1):73. doi: 10.1186/s40249-025-01322-8. Infect Dis Poverty. 2025. PMID: 40691807 Free PMC article. Review.
-
Impact of COVID-19 on Pneumococcal Acute Otitis Media, Antibiotic Resistance, and Vaccination in Children.Infect Drug Resist. 2024 Dec 13;17:5567-5578. doi: 10.2147/IDR.S496057. eCollection 2024. Infect Drug Resist. 2024. PMID: 39691490 Free PMC article. Review.
-
Effect of glucocorticoid combined with azithromycin on serum inflammatory factors and pulmonary function in children with influenza A virus-induced pneumonia.Medicine (Baltimore). 2025 Jun 6;104(23):e42117. doi: 10.1097/MD.0000000000042117. Medicine (Baltimore). 2025. PMID: 40489830 Free PMC article.
-
Prolonged mass azithromycin distributions and macrolide resistance determinants among preschool children in Niger: A sub-study of a cluster-randomized trial (MORDOR).PLoS Med. 2024 May 6;21(5):e1004386. doi: 10.1371/journal.pmed.1004386. eCollection 2024 May. PLoS Med. 2024. PMID: 38709718 Free PMC article. Clinical Trial.
References
-
- Amza A. et al. , 2017. A cluster-randomized trial to assess the efficacy of targeting trachoma treatment to children. Clin Infect Dis 64: 743–750. - PubMed

