Unusual herpetic reactivation in a young female following botox injection: a case report study
- PMID: 37784014
- PMCID: PMC10546652
- DOI: 10.1186/s12879-023-08514-3
Unusual herpetic reactivation in a young female following botox injection: a case report study
Abstract
Background: Botox injections are commonly used for cosmetic and therapeutic purposes because they temporarily paralyze muscles, reduce wrinkles, and alleviate certain medical conditions. Although generally considered safe and effective, Botox injections may cause potential complications. While herpes reactivation is more commonly associated with immunosuppressive therapies, such as chemotherapy or corticosteroid use, its association with Botox injection is poorly documented.
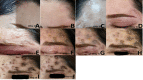
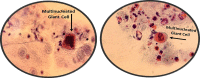
Case presentation: A 33-year-old woman presented with progressive painful rashes and vesicles on her forehead, scalp, and right upper eyelid, accompanied by fever and malaise following a Botox injection to treat wrinkles. A positive Tzanck smear test result confirmed the diagnosis of herpes infection. The patient was treated with antiviral medication, and her symptoms gradually regressed over several days.
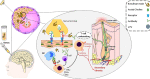
Conclusions: Although herpes reactivation is more commonly associated with immunosuppressive therapies, few cases of herpes zoster and herpes simplex following Botox injection have been reported. The pathogenesis of herpes reactivation following Botox injection is unclear; however, it has been hypothesized that the Botox protein is a potent antigen that may activate the cellular immune system, making it easier for the virus to reactivate. Healthcare providers should be aware of this potential complication and consider it when evaluating patients who present with painful rashes following Botox injections. In addition, individuals who want to receive Botox injections should be informed of this complication. The diagnosis of herpetic infection should be made promptly, and antiviral therapy should be initiated to minimize the risk of complications. Further research is needed to better understand the pathogenesis and risk factors for herpes following Botox injection and to develop strategies for preventing and managing this complication.
Keywords: Botox; Botulinum toxin; Case report; Herpes zoster; Shingles; Varicella Zoster Virus.
© 2023. BioMed Central Ltd., part of Springer Nature.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
References
-
- Ayoade F, Kumar S. Varicella Zoster (Chickenpox). StatPearls edn. Treasure Island (FL): StatPearls Publishing Copyright © 2023, StatPearls Publishing LLC.; 2023.
-
- Lopez AS, Marin M. Strategies for the control and investigation of varicella outbreaks manual, 2008. National Center for Immunization and Respiratory Diseases Center for Disease Control and Prevention, Atlanta, GA 2008.
-
- Marin M, Güris D, Chaves SS, Schmid S, Seward JF. Prevention of varicella: recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 2007;56(Rr–4):1–40. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

