Reduction of double-strand DNA break repair exacerbates vascular aging
- PMID: 37787989
- PMCID: PMC10599741
- DOI: 10.18632/aging.205066
Reduction of double-strand DNA break repair exacerbates vascular aging
Abstract
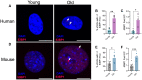
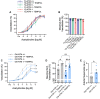
Advanced age is the greatest risk factor for cardiovascular disease (CVD), the leading cause of death. Arterial function is impaired in advanced age which contributes to the development of CVD. One underexplored hypothesis is that DNA damage within arteries leads to this dysfunction, yet evidence demonstrating the incidence and physiological consequences of DNA damage in arteries, and in particular, in the microvasculature, in advanced age is limited. In the present study, we began by assessing the abundance of DNA damage in human and mouse lung microvascular endothelial cells and found that aging increases the percentage of cells with DNA damage. To explore the physiological consequences of increases in arterial DNA damage, we evaluated measures of endothelial function, microvascular and glycocalyx properties, and arterial stiffness in mice that were lacking or heterozygous for the double-strand DNA break repair protein ATM kinase. Surprisingly, in young mice, vascular function remained unchanged which led us to rationalize that perhaps aging is required to accumulate DNA damage. Indeed, in comparison to wild type littermate controls, mice heterozygous for ATM that were aged to ~18 mo (Old ATM +/-) displayed an accelerated vascular aging phenotype characterized by increases in arterial DNA damage, senescence signaling, and impairments in endothelium-dependent dilation due to elevated oxidative stress. Furthermore, old ATM +/- mice had reduced microvascular density and glycocalyx thickness as well as increased arterial stiffness. Collectively, these data demonstrate that DNA damage that accumulates in arteries in advanced age contributes to arterial dysfunction that is known to drive CVD.
Keywords: DNA damage; aging; arterial stiffness; endothelial cell; oxidative stress; senescence; vascular function.
Conflict of interest statement
Figures



References
-
- Virani SS, Alonso A, Aparicio HJ, Benjamin EJ, Bittencourt MS, Callaway CW, Carson AP, Chamberlain AM, Cheng S, Delling FN, Elkind MSV, Evenson KR, Ferguson JF, et al. , and American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart Disease and Stroke Statistics-2021 Update: A Report From the American Heart Association. Circulation. 2021; 143:e254–743. 10.1161/CIR.0000000000000950 - DOI - PubMed
-
- World Health Organization. The top 10 causes of death World Health Organization. 2022. https://www.cdc.gov/nchs/fastats/leadingcauses-of-death.htm.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

