Recent Advances and Applications of Ambient Mass Spectrometry Imaging in Cancer Research: An Overview
- PMID: 37789912
- PMCID: PMC10542858
- DOI: 10.5702/massspectrometry.A0129
Recent Advances and Applications of Ambient Mass Spectrometry Imaging in Cancer Research: An Overview
Abstract
Cancer metabolic variability has a significant impact on both diagnosis and treatment outcomes. The discovery of novel biological indicators and metabolic dysregulation, can significantly rely on comprehension of the modified metabolism in cancer, is a research focus. Tissue histology is a critical feature in the diagnostic testing of many ailments, such as cancer. To assess the surgical margin of the tumour on patients, frozen section histology is a tedious, laborious, and typically arbitrary method. Concurrent monitoring of ion images in tissues facilitated by the latest advancements in mass spectrometry imaging (MSI) is far more efficient than optical tissue image analysis utilized in conventional histopathology examination. This article focuses on the "desorption electrospray ionization (DESI)-MSI" technique's most recent advancements and uses in cancer research. DESI-MSI can provide wealthy information based on the variances in metabolites and lipids in normal and cancerous tissues by acquiring ion images of the lipid and metabolite variances on biopsy samples. As opposed to a systematic review, this article offers a synopsis of the most widely employed cutting-edge DESI-MSI techniques in cancer research.
Keywords: DESI-MSI; ambient mass spectrometry; cancer studies; carcinoma; mass spectrometry imaging.
Copyright @ 2023 Bharath S. Kumar.
Conflict of interest statement
The authors declare no potential conflicts of interest.
Figures
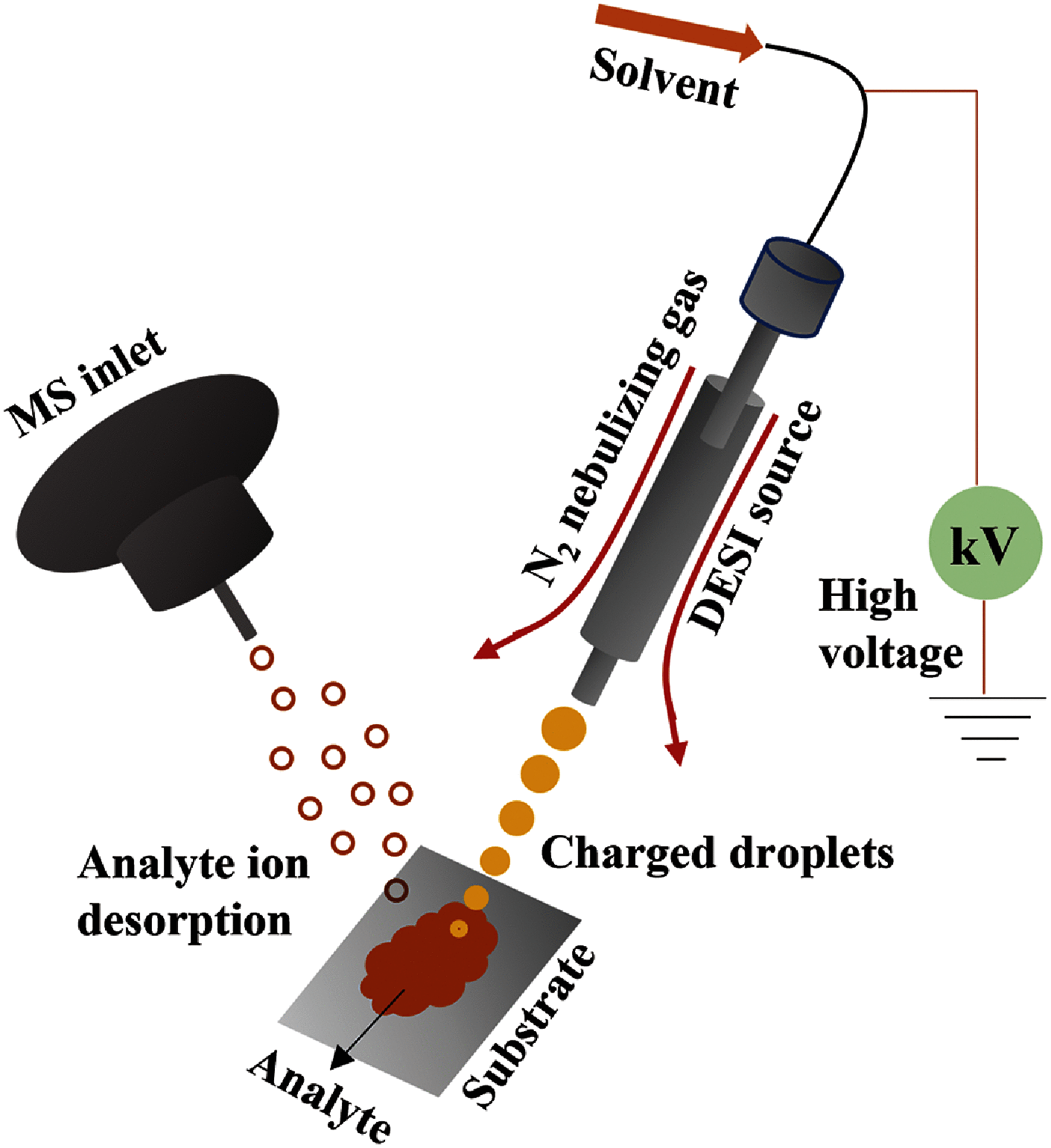

Similar articles
-
Desorption electrospray ionization mass spectrometry imaging (DESI-MSI) in disease diagnosis: an overview.Anal Methods. 2023 Aug 10;15(31):3768-3784. doi: 10.1039/d3ay00867c. Anal Methods. 2023. PMID: 37503728 Review.
-
Ambient ionization mass spectrometry imaging for disease diagnosis: Excitements and challenges.J Biosci. 2018 Sep;43(4):731-738. J Biosci. 2018. PMID: 30207318 Review.
-
Molecular characterization of human peripheral nerves using desorption electrospray ionization mass spectrometry imaging.J Anat. 2023 Nov;243(5):758-769. doi: 10.1111/joa.13909. Epub 2023 Jun 1. J Anat. 2023. PMID: 37264225 Free PMC article.
-
Lipid Coverage in Nanospray Desorption Electrospray Ionization Mass Spectrometry Imaging of Mouse Lung Tissues.Anal Chem. 2019 Sep 17;91(18):11629-11635. doi: 10.1021/acs.analchem.9b02045. Epub 2019 Aug 27. Anal Chem. 2019. PMID: 31412198 Free PMC article.
-
Mass spectrometry imaging of diclofenac and its metabolites in tissues using nanospray desorption electrospray ionization.Anal Chim Acta. 2022 Nov 15;1233:340490. doi: 10.1016/j.aca.2022.340490. Epub 2022 Oct 12. Anal Chim Acta. 2022. PMID: 36283780
Cited by
-
Assessment and interpretation of the status of surgical margins following resective surgery for head and neck squamous cancer: a narrative review.World J Surg Oncol. 2025 Aug 26;23(1):321. doi: 10.1186/s12957-025-03993-x. World J Surg Oncol. 2025. PMID: 40859320 Free PMC article. Review.
-
Breast tumor segmentation using neural cellular automata and shape guided segmentation in mammography images.PLoS One. 2024 Oct 1;19(10):e0309421. doi: 10.1371/journal.pone.0309421. eCollection 2024. PLoS One. 2024. PMID: 39352900 Free PMC article.
-
Recent Developments and Application of Mass Spectrometry Imaging in N-Glycosylation Studies: An Overview.Mass Spectrom (Tokyo). 2024;13(1):A0142. doi: 10.5702/massspectrometry.A0142. Epub 2024 Feb 27. Mass Spectrom (Tokyo). 2024. PMID: 38435075 Free PMC article. Review.
-
The burgeoning spatial multi-omics in human gastrointestinal cancers.PeerJ. 2024 Sep 13;12:e17860. doi: 10.7717/peerj.17860. eCollection 2024. PeerJ. 2024. PMID: 39285924 Free PMC article. Review.
References
-
- N. McGranahan, C. Swanton. Clonal heterogeneity and tumor evolution: Past, present, and the future. Cell 168: 613–628, 2017. - PubMed
-
- A. M. Lewandowska, M. Rudzki, S. Rudzki, T. Lewandowski, B. Laskowska. Environmental risk factors for cancer—Review paper. Ann. Agric. Environ. Med. 26: 1–7, 2019. - PubMed
-
- K. Rojas, A. Stuckey. Breast cancer epidemiology and risk factors. Clin. Obstet. Gynecol. 59: 651–672, 2016. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials

