This is a preprint.
Protein restriction slows the development and progression of Alzheimer's disease in mice
- PMID: 37790423
- PMCID: PMC10543316
- DOI: 10.21203/rs.3.rs-3342413/v2
Protein restriction slows the development and progression of Alzheimer's disease in mice
Update in
-
Protein restriction slows the development and progression of pathology in a mouse model of Alzheimer's disease.Nat Commun. 2024 Jun 18;15(1):5217. doi: 10.1038/s41467-024-49589-z. Nat Commun. 2024. PMID: 38890307 Free PMC article.
Abstract
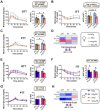
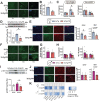
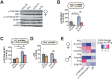
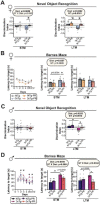
Dietary protein is a critical regulator of metabolic health and aging. Low protein diets are associated with healthy aging in humans, and many independent groups of researchers have shown that dietary protein restriction (PR) extends the lifespan and healthspan of mice. Here, we examined the effect of PR on metabolic health and the development and progression of Alzheimer's disease (AD) in the 3xTg mouse model of AD. We found that PR has metabolic benefits for 3xTg mice and non-transgenic controls of both sexes, promoting leanness and glycemic control in 3xTg mice and rescuing the glucose intolerance of 3xTg females. We found that PR induces sex-specific alterations in circulating metabolites and in the brain metabolome and lipidome, downregulating sphingolipid subclasses including ceramides, glucosylceramides, and sphingomyelins in 3xTg females. Consumption of a PR diet starting at 6 months of age reduced AD pathology in conjunction with reduced mTORC1 activity, increased autophagy, and had cognitive benefits for 3xTg mice. Finally, PR improved the survival of 3xTg mice. Our results demonstrate that PR slows the progression of AD at molecular and pathological levels, preserves cognition in this mouse model of AD, and suggests that PR or pharmaceutical interventions that mimic the effects of this diet may hold promise as a treatment for AD.
Keywords: 3xTg; Alzheimer’s disease; autophagy; mTORC1; protein restriction.
Conflict of interest statement
DWL has received funding from, and is a scientific advisory board member of, Aeovian Pharmaceuticals, which seeks to develop novel, selective mTOR inhibitors for the treatment of various diseases. J.M.D. is a consultant for Evrys Bio and co-founder of Galilei BioSciences.
Figures





References
Publication types
Grants and funding
- R56 AG056771/AG/NIA NIH HHS/United States
- F31 AG066311/AG/NIA NIH HHS/United States
- I01 BX004031/BX/BLRD VA/United States
- R01 AG084156/AG/NIA NIH HHS/United States
- R21 AG061635/AG/NIA NIH HHS/United States
- R01 AG062328/AG/NIA NIH HHS/United States
- F32 AG077916/AG/NIA NIH HHS/United States
- T32 AG000213/AG/NIA NIH HHS/United States
- RF1 AG056771/AG/NIA NIH HHS/United States
- R35 GM149279/GM/NIGMS NIH HHS/United States
- P30 DK020579/DK/NIDDK NIH HHS/United States
- R01 AG078794/AG/NIA NIH HHS/United States
- U01 AG081482/AG/NIA NIH HHS/United States
- F31 AG081115/AG/NIA NIH HHS/United States
- R01 DK133479/DK/NIDDK NIH HHS/United States
- IS1 BX005524/BX/BLRD VA/United States
- R01 AG056771/AG/NIA NIH HHS/United States
- F31 AG082504/AG/NIA NIH HHS/United States
- R35 GM151130/GM/NIGMS NIH HHS/United States
- R01 GM148487/GM/NIGMS NIH HHS/United States
- R01 NS094154/NS/NINDS NIH HHS/United States
- R01 DK125859/DK/NIDDK NIH HHS/United States
- K12 HD101368/HD/NICHD NIH HHS/United States
LinkOut - more resources
Full Text Sources
Research Materials

