Monitoring metrics over time: Why clinical trialists need to systematically collect site performance metrics
- PMID: 37795045
- PMCID: PMC7615148
- DOI: 10.1177/26320843221147855
Monitoring metrics over time: Why clinical trialists need to systematically collect site performance metrics
Abstract
Background: Over the last decade, there has been an increasing interest in risk-based monitoring (RBM) in clinical trials, resulting in a number of guidelines from regulators and its inclusion in ICH GCP. However, there is a lack of detail on how to approach RBM from a practical perspective, and insufficient understanding of best practice.
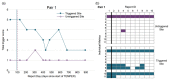
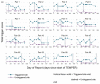
Purpose: We present a method for clinical trials units to track their metrics within clinical trials using descriptive statistics and visualisations.
Research design: We suggest descriptive statistics and visualisations within a SWAT methodology.
Study sample: We illustrate this method using the metrics from TEMPER, a monitoring study carried out in three trials at the MRC Clinical Trials Unit at UCL.
Data collection: The data collection for TEMPER is described in DOI: 10.1177/1740774518793379.
Results: We show the results and discuss a protocol for a Study-Within-A-Trial (SWAT 167) for those wishing to use the method.
Conclusions: The potential benefits metric tracking brings to clinical trials include enhanced assessment of sites for potential corrective action, improved evaluation and contextualisation of the influence of metrics and their thresholds, and the establishment of best practice in RBM. The standardisation of the collection of such monitoring data would benefit both individual trials and the clinical trials community.
Keywords: Risk-based monitoring; Study-Within-A-Trial (SWAT); central monitoring; centralised monitoring; clinical trials.
Conflict of interest statement
Declaration of conflicting interests The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors have completed the ICMJE uniform disclosure form at www.icmje.org/downloads/coi_disclosure.docx. MRS reports grants and non-financial support from Astellas, grants from Clovis Oncology, grants and non-financial support from Janssen, grants and non-financial support from Novartis, grants and non-financial support from Pfizer, grants and non-financial support from Sanofi, outside the submitted work. He also reports honoraria from Lilly Oncology and honoraria from Janssen, outside the submitted work, for clinical trial education lectures, the content of which was chosen only by the speaker. MLM holds an honorary position for the NHS DigiTrials Programme, outside the submitted work.
Figures





References
-
- European Medicines Agency. Reflection paper on risk based quality management in clinical trials EMA/269011/2013. 2013. [accessed 8 October 2021]. https://www.ema.europa.eu/en/documents/scientific-guideline/reflection-p... .
-
- FDA. Guidance to industry. Oversight of clinical investigations-A risk-based approach to monitoring. FDA-2011-D-0597. 2013. [accessed 8 October 2021]. https://www.fda.gov/regulatory-information/search-fda-guidance-documents... .
-
- International Conference on Harmonisation of technical requirements for pharmaceuticals for human use (ICH). Guideline for good clinical practice E6(R2) 2018. [accessed 8 October 2021]. https://www.ema.europa.eu/en/documents/scientific-guideline/ich-e-6-r2-g... .
-
- Fox L, Love S, Fitt L, et al. UKCRC Registered CTU Network – monitoring of clinical trials: a handbook. 2021. [accessed 14 October 2022]. https://ctru.leeds.ac.uk/download/4349/
Grants and funding
LinkOut - more resources
Full Text Sources
