Helicobacter pylori infection increases the risk of carotid plaque formation: Clinical samples combined with bioinformatics analysis
- PMID: 37809782
- PMCID: PMC10559771
- DOI: 10.1016/j.heliyon.2023.e20037
Helicobacter pylori infection increases the risk of carotid plaque formation: Clinical samples combined with bioinformatics analysis
Abstract
Objective: Infection with Helicobacter pylori (H. pylori) may increase atherosclerosis, which can lead to carotid plaque formation. Our study examined the relationship between H. pylori infection and carotid plaque formation, and its underlying mechanisms.
Methods: A total of 36,470 people who underwent physical examination in Taizhou Hospital Health Examination Center from June 2017 to June 2022 were included in this study. All people participated in the urease test, neck ultrasound, blood pressure detection, anthropometric measurement and biochemical laboratory examination. In addition, the GSE27411 and GSE28829 datasets in the Gene Expression Omnibus (GEO) database were used to analyze the mechanism of H. pylori infection and atherosclerosis progression.
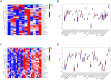
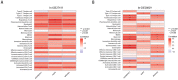
Results: H. pylori infection, sex, age, blood lipids, blood pressure, fasting blood glucose, glycated hemoglobin and body mass index were risk factors for carotid plaque formation. An independent risk factor was still evident in the multivariate logistic regression analysis, indicating H. pylori infection. Furthermore, after weighted gene coexpression network analysis (WGCNA), we discovered 555 genes linked to both H. pylori infection and the advancement of atherosclerosis. Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analyses revealed a strong correlation between these genes and immunity, infection, and immune disorders. SsGSEA analysis showed that H. pylori infection and atherosclerosis included changes in the immune microenvironment. Finally, three genes MS4A6A, ADAMDEC1 and AQP9 were identified to be involved in the formation of atherosclerosis after H. pylori infection. Conclusion: Our research affirms that H. pylori is a unique contributor to the formation of carotid plaque, examines the immune microenvironment associated with H. pylori infection and advanced carotid atherosclerosis, and offers fresh perspectives on how H. pylori infection leads to atherosclerosis.
Keywords: Bioinformatics analysis; Carotid plaque; Gene expression; Helicobacter pylori; Immune microenvironment.
© 2023 The Authors.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures




References
LinkOut - more resources
Full Text Sources
Miscellaneous

