Gold(I)-Catalyzed 1,6-Enyne Single-Cleavage Rearrangements: The Complete Picture
- PMID: 37810414
- PMCID: PMC10557124
- DOI: 10.1021/acsorginorgau.3c00028
Gold(I)-Catalyzed 1,6-Enyne Single-Cleavage Rearrangements: The Complete Picture
Abstract
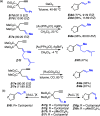
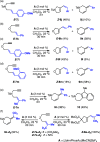
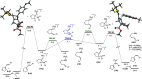
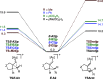
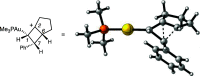
We identify the factors that rule the selectivity in single-cleavage skeletal rearrangements promoted by gold(I) catalysts. We find that stereoconvergence is enabled by a rotational equilibrium when electron-rich substituents are used. The anomalous Z-selective skeletal rearrangement is found to be due to electronic factors, whereas endo-selectivity depends on both steric and electronic factors.
© 2023 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures


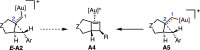

Similar articles
-
Gold(I) Complexes Nuclearity in Constrained Ferrocenyl Diphosphines: Dramatic Effect in Gold-Catalyzed Enyne Cycloisomerization.Chem Asian J. 2020 Sep 15;15(18):2879-2885. doi: 10.1002/asia.202000579. Epub 2020 Aug 10. Chem Asian J. 2020. PMID: 32687260
-
Gold(I)-catalyzed cyclizations of 1,6-enynes: alkoxycyclizations and exo/endo skeletal rearrangements.Chemistry. 2006 Feb 8;12(6):1677-93. doi: 10.1002/chem.200501088. Chemistry. 2006. PMID: 16358351
-
The role of cyclobutenes in gold(I)-catalysed skeletal rearrangement of 1,6-enynes.Org Biomol Chem. 2012 Aug 14;10(30):6105-11. doi: 10.1039/c2ob25419k. Epub 2012 May 31. Org Biomol Chem. 2012. PMID: 22648404
-
Nanocatalysts for High Selectivity Enyne Cyclization: Oxidative Surface Reorganization of Gold Sub-2-nm Nanoparticle Networks.JACS Au. 2021 Jan 25;1(2):187-200. doi: 10.1021/jacsau.0c00062. eCollection 2021 Feb 22. JACS Au. 2021. PMID: 34467283 Free PMC article.
-
Divergent Gold(I)-Catalyzed Skeletal Rearrangements of 1,7-Enynes.Chemistry. 2015 Sep 21;21(39):13526-30. doi: 10.1002/chem.201502843. Chemistry. 2015. PMID: 26356499
References
-
- Hashmi A. S. K. Homogeneous gold catalysts and alkynes: A successful liaison. Gold Bull. 2003, 36, 3–9. 10.1007/BF03214859. - DOI
LinkOut - more resources
Full Text Sources
