Catalytic 4-exo-dig carbocyclization for the construction of furan-fused cyclobutanones and synthetic applications
- PMID: 37821471
- PMCID: PMC10567718
- DOI: 10.1038/s41467-023-42032-9
Catalytic 4-exo-dig carbocyclization for the construction of furan-fused cyclobutanones and synthetic applications
Abstract
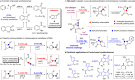
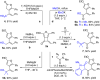
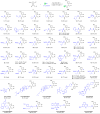
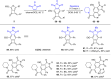
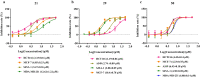
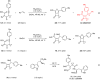
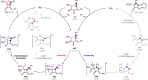
Cyclobutanone is a strained motif with broad applications, while direct assembly of the aromatic ring fused cyclobutanones beyond benzocyclobutenone (BCB) skeletons remains challenging. Herein, we report a Rh-catalyzed formal [3+2] annulation of diazo group tethered alkynes involving a 4-exo-dig carbocyclization process, providing a straightforward access to furan-fused cyclobutanones. DFT calculations disclose that, by comparison to the competitive 5-endo-dig process, 4-exo-dig carbocyclization is mainly due to lower angle strain of the key sp-hybridized vinyl cationic transition state in the cyclization step. Using less reactive catalysts Rh2(carboxylate)4 is critical for high selectivity, which is explained as catalyst-substrate hydrogen bonding interaction. This method is proved successful to direct access previously inaccessible and unknown furan-fused cyclobutanone scaffolds, which can participate in a variety of post-functionalization reactions as versatile synthetic blocks. In addition, preliminary antitumor activity study of these products indicates that some molecules exhibite significant anticancer potency against different human cancer cell lines.
© 2023. Springer Nature Limited.
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Catalytic [4+2]- and [4+4]-cycloaddition using furan-fused cyclobutanone as a privileged C4 synthon.Nat Commun. 2024 Jun 26;15(1):5407. doi: 10.1038/s41467-024-49664-5. Nat Commun. 2024. PMID: 38926359 Free PMC article.
-
Gold-catalyzed carbocyclization and imidization of alkyne-tethered diazo compounds with nitrosoarenes for the synthesis of nitrones and naphthalene derivatives.Mol Divers. 2023 Oct;27(5):1971-1978. doi: 10.1007/s11030-022-10530-5. Epub 2022 Sep 23. Mol Divers. 2023. PMID: 36149543
-
Thermodynamic and strain effects in the competition between 5-exo-dig and 6-endo-dig cyclizations of vinyl and aryl radicals.J Am Chem Soc. 2005 Sep 14;127(36):12583-94. doi: 10.1021/ja052677y. J Am Chem Soc. 2005. PMID: 16144406
-
[RhCp*Cl2]2-Catalyzed Indole Functionalization: Synthesis of Bioinspired Indole-Fused Polycycles.ACS Omega. 2023 Jul 25;8(31):27894-27919. doi: 10.1021/acsomega.3c02510. eCollection 2023 Aug 8. ACS Omega. 2023. PMID: 37576617 Free PMC article. Review.
-
Palladium- and nickel-catalyzed cascade enantioselective ring-opening/coupling reactions of cyclobutanones.Chem Commun (Camb). 2023 Mar 16;59(23):3373-3382. doi: 10.1039/d3cc00205e. Chem Commun (Camb). 2023. PMID: 36806356 Review.
Cited by
-
Catalytic [4+2]- and [4+4]-cycloaddition using furan-fused cyclobutanone as a privileged C4 synthon.Nat Commun. 2024 Jun 26;15(1):5407. doi: 10.1038/s41467-024-49664-5. Nat Commun. 2024. PMID: 38926359 Free PMC article.
-
Enantioselective Assembly of Fully Substituted α-Amino Allenoates Through a Mannich Addition and Stepwise [3,3]-σ Rearrangement Sequence.Adv Sci (Weinh). 2025 Jan;12(2):e2409334. doi: 10.1002/advs.202409334. Epub 2024 Nov 20. Adv Sci (Weinh). 2025. PMID: 39568322 Free PMC article.
-
Rare Gold-Catalyzed 4-exo-dig Cyclization for Ring Expansion of Propargylic Aziridines toward Stereoselective (Z)-Alkylidene Azetidines, via Diborylalkyl Homopropargyl Amines.Org Lett. 2024 Sep 13;26(36):7535-7540. doi: 10.1021/acs.orglett.4c02415. Epub 2024 Sep 2. Org Lett. 2024. PMID: 39219538 Free PMC article.
References
-
- Fumagalli G, Stanton S, Bower JF. Recent methodologies that exploit C–C single-bond cleavage of strained ring systems by transition metal complexes. Chem. Rev. 2017;117:9404–9432. - PubMed
-
- Mack DJ, Njardarson JT. Recent advances in the metal-catalyzed ring expansions of three- and four-membered rings. ACS Catal. 2013;3:272–286.
-
- Ebner C, Carreira EM. Cyclopropanation strategies in recent total syntheses. Chem. Rev. 2017;117:11651–11679. - PubMed
-
- Wang F, Yu S, Li X. Transition metal-catalysed couplings between arenes and strained or reactive rings: combination of C–H activation and ring scission. Chem. Soc. Rev. 2016;45:6462–6477. - PubMed
-
- Beniddir MA, Evanno L, Joseph D, Skiredj A, Poupon E. Emergence of diversity and stereochemical outcomes in the biosynthetic pathways of cyclobutane-centered marine alkaloid dimers. Nat. Prod. Rep. 2016;33:820–842. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

