Mega-dose sodium ascorbate: a pilot, single-dose, physiological effect, double-blind, randomized, controlled trial
- PMID: 37828547
- PMCID: PMC10571252
- DOI: 10.1186/s13054-023-04644-x
Mega-dose sodium ascorbate: a pilot, single-dose, physiological effect, double-blind, randomized, controlled trial
Abstract
Background: Mega-dose sodium ascorbate (NaAscorbate) appears beneficial in experimental sepsis. However, its physiological effects in patients with septic shock are unknown.
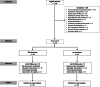
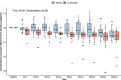
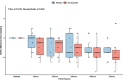
Methods: We conducted a pilot, single-dose, double-blind, randomized controlled trial. We enrolled patients with septic shock within 24 h of diagnosis. We randomly assigned them to receive a single mega-dose of NaAscorbate (30 g over 1 h followed by 30 g over 5 h) or placebo (vehicle). The primary outcome was the total 24 h urine output (UO) from the beginning of the study treatment. Secondary outcomes included the time course of the progressive cumulative UO, vasopressor dose, and sequential organ failure assessment (SOFA) score.
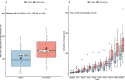
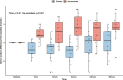
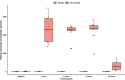
Results: We enrolled 30 patients (15 patients in each arm). The mean (95% confidence interval) total 24-h UO was 2056 (1520-2593) ml with placebo and 2948 (2181-3715) ml with NaAscorbate (mean difference 891.5, 95% confidence interval [- 2.1 to 1785.2], P = 0.051). Moreover, the progressive cumulative UO was greater over time on linear mixed modelling with NaAscorbate (P < 0.001). Vasopressor dose and SOFA score changes over time showed faster reductions with NaAscorbate (P < 0.001 and P = 0.042). The sodium level, however, increased more over time with NaAscorbate (P < 0.001). There was no statistical difference in other clinical outcomes.
Conclusion: In patients with septic shock, mega-dose NaAscorbate did not significantly increase cumulative 24-h UO. However, it induced a significantly greater increase in UO and a greater reduction in vasopressor dose and SOFA score over time. One episode of hypernatremia and one of hemolysis were observed in the NaAscorbate group. These findings support further cautious investigation of this novel intervention. Trial registration Australian New Zealand Clinical Trial Registry (ACTRN12620000651987), Date registered June/5/2020.
Keywords: Sepsis; Septic shock; Sequential organ failure score; Sodium ascorbate; Vasopressors; Vitamin C.
© 2023. BioMed Central Ltd., part of Springer Nature.
Conflict of interest statement
YRL, CNM and RB own a patent for the use of mega-dose sodium ascorbate in the treatment of sepsis.
Figures
References
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

