A Drosophila model targets Eiger/TNFα to alleviate obesity-related insulin resistance and macrophage infiltration
- PMID: 37828911
- PMCID: PMC10651092
- DOI: 10.1242/dmm.050388
A Drosophila model targets Eiger/TNFα to alleviate obesity-related insulin resistance and macrophage infiltration
Abstract
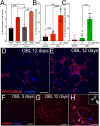
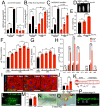
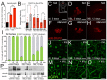
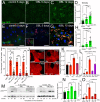
Obesity is associated with various metabolic disorders, such as insulin resistance and adipose tissue inflammation (ATM), characterized by macrophage infiltration into adipose cells. This study presents a new Drosophila model to investigate the mechanisms underlying these obesity-related pathologies. We employed genetic manipulation to reduce ecdysone levels to prolong the larval stage. These animals are hyperphagic and exhibit features resembling obesity in mammals, including increased lipid storage, adipocyte hypertrophy and high circulating glucose levels. Moreover, we observed significant infiltration of immune cells (hemocytes) into the fat bodies, accompanied by insulin resistance. We found that attenuation of Eiger/TNFα signaling reduced ATM and improved insulin sensitivity. Furthermore, using metformin and the antioxidants anthocyanins, we ameliorated both phenotypes. Our data highlight evolutionarily conserved mechanisms allowing the development of Drosophila models for discovering therapeutic pathways in adipose tissue immune cell infiltration and insulin resistance. Our model can also provide a platform to perform genetic screens or test the efficacy of therapeutic interventions for diseases such as obesity, type 2 diabetes and non-alcoholic fatty liver disease.
Keywords: Drosophila model; Adipose chronic inflammation; Eiger/TNFα signaling; Insulin resistance; Lipid metabolism; Metabolic disorders; Obesity.
© 2023. Published by The Company of Biologists Ltd.
Conflict of interest statement
Competing interests The authors declare no competing or financial interests.
Figures

Update of
-
A Novel Drosophila Model to Investigate Adipose Tissue Macrophage Infiltration (ATM) and Obesity highlights the Therapeutic Potential of Attenuating Eiger/TNFα Signaling to Ameliorate Insulin Resistance and ATM.bioRxiv [Preprint]. 2023 Jul 8:2023.07.06.548016. doi: 10.1101/2023.07.06.548016. bioRxiv. 2023. Update in: Dis Model Mech. 2023 Nov 1;16(11):dmm050388. doi: 10.1242/dmm.050388. PMID: 37461586 Free PMC article. Updated. Preprint.
References
-
- Andersen, D. S., Colombani, J., Palmerini, V., Chakrabandhu, K., Boone, E., Rothlisberger, M., Toggweiler, J., Basler, K., Mapelli, M., Hueber, A. O.et al. (2015). The Drosophila TNF receptor Grindelwald couples loss of cell polarity and neoplastic growth. Nature 522, 482-486. 10.1038/nature14298 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

