Single-nuclei characterization of pervasive transcriptional signatures across organs in response to COVID-19
- PMID: 37830426
- PMCID: PMC10575628
- DOI: 10.7554/eLife.81090
Single-nuclei characterization of pervasive transcriptional signatures across organs in response to COVID-19
Abstract
Background: Infection by coronavirus SARS-CoV2 is a severe and often deadly disease that has implications for the respiratory system and multiple organs across the human body. While the effects in the lung have been extensively studied, less is known about the impact COVID-19 has across other organs.
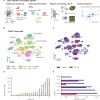
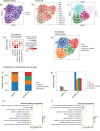
Methods: Here, we contribute a single-nuclei RNA-sequencing atlas comprising six human organs across 20 autopsies where we analyzed the transcriptional changes due to COVID-19 in multiple cell types. The integration of data from multiple organs enabled the identification of systemic transcriptional changes.
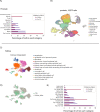
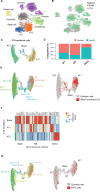
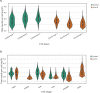
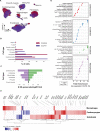
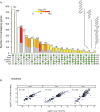
Results: Computational cross-organ analysis for endothelial cells and macrophages identified systemic transcriptional changes in these cell types in COVID-19 samples. In addition, analysis of gene modules showed enrichment of specific signaling pathways across multiple organs in COVID-19 autopsies.
Conclusions: Altogether, the COVID Tissue Atlas enables the investigation of both cell type-specific and cross-organ transcriptional responses to COVID-19, providing insights into the molecular networks affected by the disease and highlighting novel potential targets for therapies and drug development.
Funding: The Chan-Zuckerberg Initiative, The Chan-Zuckerberg Biohub.
Keywords: COVID; RNA-seq; cell atlas; epidemiology; genetics; genomics; global health; human; single-cell; transcriptional regulation; transcriptomics.
© 2023, The COVID Tissue Atlas Consortium et al.
Conflict of interest statement
AG, SB, HS, AA, AC, TP, NN, AP, FH, BW No competing interests declared
Figures








Update of
References
-
- Adams TS, Schupp JC, Poli S, Ayaub EA, Neumark N, Ahangari F, Chu SG, Raby BA, DeIuliis G, Januszyk M, Duan Q, Arnett HA, Siddiqui A, Washko GR, Homer R, Yan X, Rosas IO, Kaminski N. Single-cell RNA-seq reveals ectopic and aberrant lung-resident cell populations in idiopathic pulmonary fibrosis. Science Advances. 2020;6:eaba1983. doi: 10.1126/sciadv.aba1983. - DOI - PMC - PubMed
-
- Deinhardt-Emmer S, Wittschieber D, Sanft J, Kleemann S, Elschner S, Haupt KF, Vau V, Häring C, Rödel J, Henke A, Ehrhardt C, Bauer M, Philipp M, Gaßler N, Nietzsche S, Löffler B, Mall G. Early postmortem mapping of SARS-CoV-2 RNA in patients with COVID-19 and the correlation with tissue damage. eLife. 2021;10:e60361. doi: 10.7554/eLife.60361. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions

