Investigating trait variability of gene co-expression network architecture in brain by controlling for genomic risk of schizophrenia
- PMID: 37831723
- PMCID: PMC10599557
- DOI: 10.1371/journal.pgen.1010989
Investigating trait variability of gene co-expression network architecture in brain by controlling for genomic risk of schizophrenia
Abstract
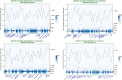
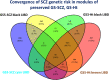
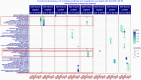
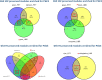
The effect of schizophrenia (SCZ) genetic risk on gene expression in brain remains elusive. A popular approach to this problem has been the application of gene co-expression network algorithms (e.g., WGCNA). To improve reliability with this method it is critical to remove unwanted sources of variance while also preserving biological signals of interest. In this WCGNA study of RNA-Seq data from postmortem prefrontal cortex (78 neurotypical donors, EUR ancestry), we tested the effects of SCZ genetic risk on co-expression networks. Specifically, we implemented a novel design in which gene expression was adjusted by linear regression models to preserve or remove variance explained by biological signal of interest (GWAS genomic scores for SCZ risk-(GS-SCZ), and genomic scores- GS of height (GS-Ht) as a negative control), while removing variance explained by covariates of non-interest. We calculated co-expression networks from adjusted expression (GS-SCZ and GS-Ht preserved or removed), and consensus between them (representative of a "background" network free of genomic scores effects). We then tested the overlap between GS-SCZ preserved modules and background networks reasoning that modules with reduced overlap would be most affected by GS-SCZ biology. Additionally, we tested these modules for convergence of SCZ risk (i.e., enrichment in PGC3 SCZ GWAS priority genes, enrichment in SCZ risk heritability and relevant biological ontologies. Our results highlight key aspects of GS-SCZ effects on brain co-expression networks, specifically: 1) preserving/removing SCZ genetic risk alters the co-expression modules; 2) biological pathways enriched in modules affected by GS-SCZ implicate processes of transcription, translation and metabolism that converge to influence synaptic transmission; 3) priority PGC3 SCZ GWAS genes and SCZ risk heritability are enriched in modules associated with GS-SCZ effects. Overall, our results indicate that gene co-expression networks that selectively integrate information about genetic risk can reveal novel combinations of biological pathways involved in schizophrenia.
Copyright: © 2023 Radulescu et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
I have read the journal’s policy and the authors of this manuscript have the following competing interests: DW serves on the Scientific Advisory Boards of Sage Therapeutics and Pasithea Therapeutics. JK is a member of an external Data Monitoring Committee (eDMC) for Merck on a clinical trial for a novel antipsychotic for patients with schizophrenia. The other authors declare no competing interests.
Figures


Similar articles
-
Identification and prioritization of gene sets associated with schizophrenia risk by co-expression network analysis in human brain.Mol Psychiatry. 2020 Apr;25(4):791-804. doi: 10.1038/s41380-018-0304-1. Epub 2018 Nov 26. Mol Psychiatry. 2020. PMID: 30478419
-
Network-wide risk convergence in gene co-expression identifies reproducible genetic hubs of schizophrenia risk.Neuron. 2024 Nov 6;112(21):3551-3566.e6. doi: 10.1016/j.neuron.2024.08.005. Epub 2024 Sep 4. Neuron. 2024. PMID: 39236717
-
Transcriptome sequencing and genome-wide association analyses reveal lysosomal function and actin cytoskeleton remodeling in schizophrenia and bipolar disorder.Mol Psychiatry. 2015 May;20(5):563-572. doi: 10.1038/mp.2014.82. Epub 2014 Aug 12. Mol Psychiatry. 2015. PMID: 25113377 Free PMC article.
-
How can genetics help understand the relationship between cognitive dysfunction and schizophrenia?Scand J Psychol. 2018 Feb;59(1):26-31. doi: 10.1111/sjop.12407. Scand J Psychol. 2018. PMID: 29356008 Review.
-
Genetic evidence for role of integration of fast and slow neurotransmission in schizophrenia.Mol Psychiatry. 2017 Jun;22(6):792-801. doi: 10.1038/mp.2017.33. Epub 2017 Mar 28. Mol Psychiatry. 2017. PMID: 28348379 Free PMC article. Review.
References
-
- Bleuler E (1950). Dementia Praecox or the Group of Schizophrenias. Zinkin J, translator. New York, NY: International Universities Press.
-
- Friston KJ, Frith CD. Schizophrenia: A disconnection syndrome? Clin Neurosci 1995; 3: 89–97. - PubMed
-
- Collado-Torres L, Burke EE, Peterson A, Shin J, Straub RE, Rajpurohit A, et al.. Regional heterogeneity in gene expression, regulation, and coherence in the frontal cortex and hippocampus across development and schizophrenia. Neuron 2019; 103: 203,216.e8. doi: 10.1016/j.neuron.2019.05.013 - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
- R01 MH109897/MH/NIMH NIH HHS/United States
- R37 MH057881/MH/NIMH NIH HHS/United States
- R01 MH075916/MH/NIMH NIH HHS/United States
- U01 MH103392/MH/NIMH NIH HHS/United States
- R01 MH109677/MH/NIMH NIH HHS/United States
- P50 MH084053/MH/NIMH NIH HHS/United States
- HHSN271201300031C/DA/NIDA NIH HHS/United States
- R01 MH110921/MH/NIMH NIH HHS/United States
- P50 MH066392/MH/NIMH NIH HHS/United States
- R01 MH080405/MH/NIMH NIH HHS/United States
- R01 MH085542/MH/NIMH NIH HHS/United States
- R01 MH097276/MH/NIMH NIH HHS/United States
- P01 AG002219/AG/NIA NIH HHS/United States
- R01 MH093725/MH/NIMH NIH HHS/United States
- P50 AG005138/AG/NIA NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical

