Use of a taurolidine containing antimicrobial wash to reduce cardiac implantable electronic device infection
- PMID: 37831737
- PMCID: PMC10616572
- DOI: 10.1093/europace/euad306
Use of a taurolidine containing antimicrobial wash to reduce cardiac implantable electronic device infection
Abstract
Aims: TauroPace (Tauropharm, Bavaria Germany), a taurolidine solution for combating cardiac implantable electronic device (CIED) infection, was compared with a historical control of 3% hydrogen peroxide (H2O2) in a prospective observational study.
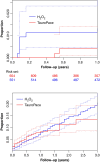
Methods and results: The device pocket was irrigated, and all hardware accessible within (leads, suture sleeves, pulse generator) was wiped with H2O2, TauroPace, or taurolidine in a galenic formulation during any invasive CIED procedure at the study centre. Only CIED procedures covered by TauroPace or H2O2 from 1 January 2017 to 28 February 2022 were included for analysis. Patients who underwent >1 procedure were censored for the last treatment group and reassigned at the next procedure. The primary endpoint was major CIED infection within 3 months. The secondary endpoints were CIED infection beyond 3 months, adverse events potentially related to the antimicrobial solutions, CIED system, procedure, and death, till the end of follow-up. TauroPace covered 654 procedures on 631 patients, and H2O2 covered 551 procedures on 532 patients. The TauroPace group had more patient risk factors for infection than the H2O2 group (P = 0.0058) but similar device and procedure-specific risk factors (P = 0.17). Cardiac implantable electronic device infection occurred in 0/654 (0%) of the TauroPace group and 6/551 (1.1%) of the H2O2 group (P = 0.0075). Death occurred in 23/654 (3.5%) of the TauroPace group and 14/551 (2.5%) of the H2O2 group (P = 0.33). Non-infection related adverse events were rarer in the TauroPace (3.8%) than the H2O2 (6.0%) group (P = 0.0802).
Conclusion: TauroPace is safe but more effective than H2O2 in reducing CIED infection.
Clinical trial registration: ClinicalTrials.gov Identifier: NCT05576194.
Keywords: Cardiac implantable electronic device; Hydrogen peroxide; Infection; Taurolidine; Tauropace.
© The Author(s) 2023. Published by Oxford University Press on behalf of the European Society of Cardiology.
Conflict of interest statement
Conflict of interest: B.B.: consultant/advisor: Abbott, Bioline Supply, Crosstec GmbH, Kapamed, Kimal PLC, M3 Medical/Ecclipse Medical, Medival SRL, Philips/Spectranetics, Sintec SRL, Tauro-Implant GmbH, Tauropharm GmbH, Transcutan; the other authors declare no conflict of interest in relation to this manuscript.
Figures

Comment in
-
Taurolidine: could this be the 'silver bullet' against cardiac implantable electronic device infection?Europace. 2023 Nov 2;25(11):euad332. doi: 10.1093/europace/euad332. Europace. 2023. PMID: 37967367 Free PMC article. No abstract available.
References
-
- Kusumoto FM, Schoenfeld MH, Wilkoff BL, Berul CI, Birgersdotter-Green UM, Carrillo Ret al. . 2017 HRS expert consensus statement on cardiovascular implantable electronic device lead management and extraction. Heart Rhythm 2017;14:e503–51. - PubMed
-
- Greenspon AJ, Patel JD, Lau E, Ochoa JA, Frisch DR, Ho RTet al. . 16-year Trends in the infection burden for pacemakers and implantable cardioverter-defibrillators in the United States 1993 to 2008. J Am Coll Cardiol 2011;58:1001–6. - PubMed
-
- Voigt A, Shalaby A, Saba S. Continued rise in rates of cardiovascular implantable electronic device infections in the United States: temporal trends and causative insights. Pacing Clin Electrophysiol 2010;33:414–9. - PubMed
-
- Poole JE, Gleva MJ, Mela T, Chung MK, Uslan DZ, Borge Ret al. . Complication rates associated with pacemaker or implantable cardioverter-defibrillator generator replacements and upgrade procedures: results from the REPLACE registry. Circulation 2010;122:1553–61. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

