Interleukin-13 Mediates Non-Steroidal Anti-Inflammatory-Drug-Induced Small Intestinal Mucosal Injury with Ulceration
- PMID: 37834420
- PMCID: PMC10573871
- DOI: 10.3390/ijms241914971
Interleukin-13 Mediates Non-Steroidal Anti-Inflammatory-Drug-Induced Small Intestinal Mucosal Injury with Ulceration
Abstract
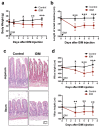
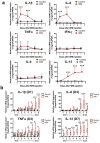
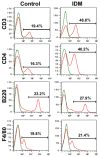
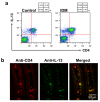
Non-steroidal anti-inflammatory drugs (NSAIDs), which are antipyretics and analgesics, cause gastrointestinal disorders, such as inflammation and ulcers. To prescribe NSAIDs more safely, it is important to clarify the mechanism of NSAID-induced gastrointestinal mucosal injury. However, there is a paucity of studies on small intestinal mucosal damage by NSAIDs, and it is currently unknown whether inflammation and ulceration also occur in the small intestine, and whether mediators are involved in the mechanism of injury. Therefore, in this study, we created an animal model in which small intestinal mucosal injury was induced using NSAIDs (indomethacin; IDM). Focusing on the dynamics of immune regulatory factors related to the injury, we aimed to elucidate the pathophysiological mechanism involved. We analyzed the pathological changes in the small intestine, the expression of immunoregulatory factors (cytokines), and identified cytokine secretion and expression cells from isolated lamina propria mononuclear cells (LPMCs). Ulcers were formed in the small intestine by administering IDM. Although the mRNA expression levels of IL-1β, IL-6, and TNFα were decreased on day 7 after IDM administration, IL-13 mRNA levels increased from day 3 after IDM administration and remained high even on day 7. The IL-13 mRNA expression and the secretion of IL-13 were increased in small intestinal LPMCs isolated from the IDM-treated group. In addition, we confirmed that IL-13 was expressed in CD4-positive T cells. These results provided new evidence that IL-13 production from CD4-positive T cells in the lamina propria of the small intestine contributes to NSAID-induced mucosal injury.
Keywords: IL-13 (Interleukin 13); LPMCs (lamina propria mononuclear cells); NSAIDs (non-steroidal anti-inflammatory drugs); small intestine; ulceration.
Conflict of interest statement
None of the authors have any conflicts of interest to declare.
Figures




Similar articles
-
Gastrointestinal mucosal injury following repeated daily oral administration of conventional formulations of indometacin and other non-steroidal anti-inflammatory drugs to pigs: a model for human gastrointestinal disease.J Pharm Pharmacol. 2003 May;55(5):661-8. doi: 10.1211/002235703765344577. J Pharm Pharmacol. 2003. PMID: 12831509
-
Preventive effect of agaro-oligosaccharides on non-steroidal anti-inflammatory drug-induced small intestinal injury in mice.J Gastroenterol Hepatol. 2014 Feb;29(2):310-7. doi: 10.1111/jgh.12373. J Gastroenterol Hepatol. 2014. PMID: 23980531
-
Down-regulation of hepatic CYP3A1 expression in a rat model of indomethacin-induced small intestinal ulcers.Biopharm Drug Dispos. 2016 Dec;37(9):522-532. doi: 10.1002/bdd.2042. Epub 2016 Oct 14. Biopharm Drug Dispos. 2016. PMID: 27666336
-
Present status and strategy of NSAIDs-induced small bowel injury.J Gastroenterol. 2009;44(9):879-88. doi: 10.1007/s00535-009-0102-2. Epub 2009 Jul 1. J Gastroenterol. 2009. PMID: 19568687 Review.
-
Non-steroidal anti-inflammatory drug gastropathy: causes and treatment.Scand J Gastroenterol Suppl. 1996;220:124-7. doi: 10.3109/00365529609094763. Scand J Gastroenterol Suppl. 1996. PMID: 8898449 Review.
Cited by
-
Dietary zinc intake associated with stroke in American adults.Sci Rep. 2025 May 26;15(1):18301. doi: 10.1038/s41598-025-03122-4. Sci Rep. 2025. PMID: 40419632 Free PMC article.
-
Tamarix aphylla derived metabolites ameliorate indomethacin-induced gastric ulcers in rats by modulating the MAPK signaling pathway, alleviating oxidative stress and inflammation: In vivo study supported by pharmacological network analysis.PLoS One. 2024 May 10;19(5):e0302015. doi: 10.1371/journal.pone.0302015. eCollection 2024. PLoS One. 2024. PMID: 38728332 Free PMC article.
-
Small Intestinal Ulceration in Two Recurrence-Free Young Patients at 10 Years Postoperatively.Cureus. 2025 Jul 13;17(7):e87856. doi: 10.7759/cureus.87856. eCollection 2025 Jul. Cureus. 2025. PMID: 40799892 Free PMC article.
References
-
- Fabijanić D., Kardum D., Banić M., Fabijanić A. Nonsteroidal-anti-inflammatory drugs and serious undesirable gastrointestinal effects. Lijec. Vjesn. 2007;129:205–213. - PubMed
-
- Singh G., Triadafilopoulos G. Epidemiology of NSAID induced gastrointestinal complications. J. Rheumatol. Suppl. 1999;56:18–24. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

