Updates on Emerging Interventions for Autosomal Recessive ABCA4-Associated Stargardt Disease
- PMID: 37834872
- PMCID: PMC10573680
- DOI: 10.3390/jcm12196229
Updates on Emerging Interventions for Autosomal Recessive ABCA4-Associated Stargardt Disease
Abstract
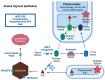
Autosomal recessive Stargardt disease (STGD1) is an inherited retinal degenerative disease associated with a mutated ATP-binding cassette, subfamily A, member 4 (ABCA4) gene. STGD1 is the most common form of juvenile macular degeneration with onset in late childhood to early or middle adulthood and causes progressive, irreversible visual impairment and blindness. No effective treatment is currently available. In the present article, we review the most recent updates in clinical trials targeting the management of STGD1, including gene therapy, small molecule therapy, and stem cell therapy. In gene therapy, dual adeno-associated virus and non-viral vectors have been successful in delivering the human ABCA4 gene in preclinical studies. For pharmaceutical therapies ALK-001, deuterated vitamin A shows promise with preliminary data for phase 2 trial, demonstrating a decreased atrophy growth rate after two years. Stem cell therapy using human pluripotent stem cell-derived retinal pigment epithelium cells demonstrated long-term safety three years after implantation and visual acuity improvements in the first two years after initiation of therapy. Many other treatment options have ongoing investigations and clinical trials. While multiple potential interventions have shown promise in attenuating disease progression, further exploration is necessary to demonstrate treatment safety and efficacy.
Keywords: ABCA4; Stargardt disease; adeno-associated viral vectors; deuterated vitamin A; emixustat; gene therapy; human embryonic stem cells; stem cell therapy; visual cycle modulators.
Conflict of interest statement
Ninel Z. Gregori receives research support from Ocugen, Endogena, Nanoscope, Gyroscope, and Biogen as well as consultation fees from Gyroscope, Novartis, BlueRock Therapeutics, and Bionic Vision Technologies. The remaining authors declare no conflicts of interest.
Figures

References
-
- Li C.H.Z., Pas J., Corradi Z., Hitti-Malin R.J., Hoogstede A., Runhart E.H., Dhooge P.P.A., Collin R.W.J., Cremers F.P.M., Hoyng C.B. Study of Late-Onset Stargardt Type 1 Disease: Characteristics, Genetics and Progression. Ophthalmology. 2023 doi: 10.1016/j.ophtha.2023.08.011. in press . - DOI - PubMed
-
- Blacharski P., Newsome D. Retinal Dystrophies and Degenerations. Raven Press; New York, NY, USA: 1988. pp. 135–159.
-
- Strauss R.W., Ho A., Muñoz B., Cideciyan A.V., Sahel J.A., Sunness J.S., Birch D.G., Bernstein P.S., Michaelides M., Traboulsi E.I., et al. The Natural History of the Progression of Atrophy Secondary to Stargardt Disease (ProgStar) Studies: Design and Baseline Characteristics: ProgStar Report No. 1. Ophthalmology. 2016;123:817–828. doi: 10.1016/j.ophtha.2015.12.009. - DOI - PubMed
-
- Fujinami K., Sergouniotis P.I., Davidson A.E., Wright G., Chana R.K., Tsunoda K., Tsubota K., Egan C.A., Robson A.G., Moore A.T., et al. Clinical and molecular analysis of Stargardt disease with preserved foveal structure and function. Am. J. Ophthalmol. 2013;156:487–501.e481. doi: 10.1016/j.ajo.2013.05.003. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

