The Value of Electroretinography in Identifying Candidate Genes for Inherited Retinal Dystrophies: A Diagnostic Guide
- PMID: 37835784
- PMCID: PMC10572658
- DOI: 10.3390/diagnostics13193041
The Value of Electroretinography in Identifying Candidate Genes for Inherited Retinal Dystrophies: A Diagnostic Guide
Abstract
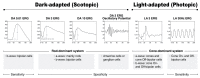
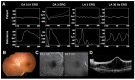
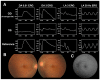
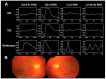
Inherited retinal dystrophies (IRDs) are a group of heterogeneous diseases caused by genetic mutations that specifically affect the function of the rod, cone, or bipolar cells in the retina. Electroretinography (ERG) is a diagnostic tool that measures the electrical activity of the retina in response to light stimuli, and it can help to determine the function of these cells. A normal ERG response consists of two waves, the a-wave and the b-wave, which reflect the activity of the photoreceptor cells and the bipolar and Muller cells, respectively. Despite the growing availability of next-generation sequencing (NGS) technology, identifying the precise genetic mutation causing an IRD can be challenging and costly. However, certain types of IRDs present with unique ERG features that can help guide genetic testing. By combining these ERG findings with other clinical information, such as on family history and retinal imaging, physicians can effectively narrow down the list of candidate genes to be sequenced, thereby reducing the cost of genetic testing. This review article focuses on certain types of IRDs with unique ERG features. We will discuss the pathophysiology and clinical presentation of, and ERG findings on, these disorders, emphasizing the unique role ERG plays in their diagnosis and genetic testing.
Keywords: X-linked retinoschisis; cone dystrophy with supernormal rod response; cone–rod dystrophies; congenital stationary night blindness; electronegative ERG; electroretinography; enhanced S-cone syndrome; fundus albipunctatus; inherited retinal dystrophies.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analysis, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures





References
-
- National Human Genome Research Institute The Cost of Sequencing a Human Genome. [(accessed on 28 May 2023)]; Available online: https://www.genome.gov/about-genomics/fact-sheets/Sequencing-Human-Genom....
-
- Purves D., Augustine G.J., Fitzpatrick D. Anatomical Distribution of Rods and Cones. 2nd ed. Sinauer Associates; Sunderland, MA, USA: 2001. Neuroscience.
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

