Studying the Degradation of Three Polymers under Different Chlorine Concentrations and Exposure Times
- PMID: 37835981
- PMCID: PMC10574910
- DOI: 10.3390/polym15193931
Studying the Degradation of Three Polymers under Different Chlorine Concentrations and Exposure Times
Abstract
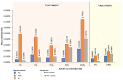
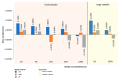
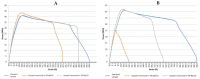
Due to chlorine's ability to kill bacteria and fungi through a chemical reaction, chlorine solutions are commonly used to clean and disinfect numerous public facilities, although these actions are also dependent to the equipment present in those facilities. Accordingly, the interest in studying its effect when in contact with different materials is obvious. This study was carried out through accelerated degradation tests and various analysis methods (optical microscope, scanning electron microscope, and tensile tests). The objective was to observe the wear presented by three polymeric materials, polyvinyl chloride (PVC), high-density polyethylene (HDPE), and polypropylene (PP), when exposed to chlorine's action in swimming pools and drinking water treatment plants. The resulting effect depends on the chlorine content and the type of contact between the chemical agent and the material. The aim was to select the material less likely to be affected by chlorine through tests and analyses, allowing a longer component life. The use of certain more resistant polymeric materials can drastically reduce maintenance, reducing fundamental factors such as costs, the downtime of municipal facilities, and also the risk to public health. It was concluded that PVC has the most stable behaviour overall when in contact with chlorine solutions.
Keywords: HDPE; PP; PVC; chlorine action; municipal facilities; polymeric degradation; polymers.
Conflict of interest statement
The authors declare no conflict of interest.
Figures















References
-
- PubChem—National Center for Biotechnology Information Element Summary, Chlorine. [(accessed on 15 May 2023)]; Available online: https://pubchem.ncbi.nlm.nih.gov/element/Chlorine.
-
- Cloro-Infopédia. [(accessed on 9 October 2022)]. Available online: https://www.infopedia.pt/apoio/artigos/$cloro.
-
- Foster A.R., Stark E.R., Ikner L.A., Pepper I.L. Bench scale investigation of the effects of a magnetic water treatment device in pool systems on chlorine demand. J. Water Process Eng. 2022;50:103198. doi: 10.1016/j.jwpe.2022.103198. - DOI
-
- Fisher I., Kastl G., Sathasivan A., Catling R. Modelling chlorine residual and trihalomethane profiles in water distribution systems after treatment including pre-chlorination. J. Environ. Chem. Eng. 2021;9:105686. doi: 10.1016/j.jece.2021.105686. - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous

