Role of plastids and mitochondria in the early development of seedlings in dark growth conditions
- PMID: 37841629
- PMCID: PMC10570830
- DOI: 10.3389/fpls.2023.1272822
Role of plastids and mitochondria in the early development of seedlings in dark growth conditions
Abstract
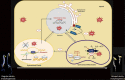
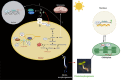
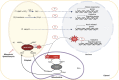
Establishment of the seedlings is a crucial stage of the plant life cycle. The success of this process is essential for the growth of the mature plant. In Nature, when seeds germinate under the soil, seedlings follow a dark-specific program called skotomorphogenesis, which is characterized by small, non-green cotyledons, long hypocotyl, and an apical hook-protecting meristematic cells. These developmental structures are required for the seedlings to emerge quickly and safely through the soil and gain autotrophy before the complete depletion of seed resources. Due to the lack of photosynthesis during this period, the seed nutrient stocks are the primary energy source for seedling development. The energy is provided by the bioenergetic organelles, mitochondria, and etioplast (plastid in the dark), to the cell in the form of ATP through mitochondrial respiration and etio-respiration processes, respectively. Recent studies suggest that the limitation of the plastidial or mitochondrial gene expression induces a drastic reprogramming of the seedling morphology in the dark. Here, we discuss the dark signaling mechanisms involved during a regular skotomorphogenesis and how the dysfunction of the bioenergetic organelles is perceived by the nucleus leading to developmental changes. We also describe the probable involvement of several plastid retrograde pathways and the interconnection between plastid and mitochondria during seedling development. Understanding the integration mechanisms of organellar signals in the developmental program of seedlings can be utilized in the future for better emergence of crops through the soil.
Keywords: development; etioplasts; mitochondria; retrograde control; skotomorphogenesis.
Copyright © 2023 Sajib, Kandel, Prity, Oukacine, Gakière and Merendino.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
Publication types
LinkOut - more resources
Full Text Sources

