Mimicking lipolytic, adipogenic, and secretory capacities of human subcutaneous adipose tissue by spheroids from distinct subpopulations of adipose stromal/stem cells
- PMID: 37842092
- PMCID: PMC10570415
- DOI: 10.3389/fcell.2023.1219218
Mimicking lipolytic, adipogenic, and secretory capacities of human subcutaneous adipose tissue by spheroids from distinct subpopulations of adipose stromal/stem cells
Abstract
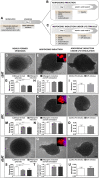
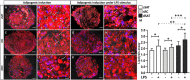
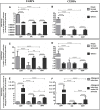
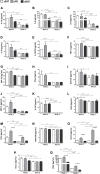
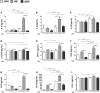
Background: Adipose tissue engineering may provide 3D models for the understanding of diseases such as obesity and type II diabetes. Recently, distinct adipose stem/stromal cell (ASC) subpopulations were identified from subcutaneous adipose tissue (SAT): superficial (sSAT), deep (dSAT), and the superficial retinacula cutis (sRC). This study aimed to test these subpopulations ASCs in 3D spheroid culture induced for adipogenesis under a pro-inflammatory stimulus with lipopolysaccharide (LPS). Methods: The samples of abdominal human subcutaneous adipose tissue were obtained during plastic aesthetic surgery (Protocol 145/09). Results: ASC spheroids showed high response to adipogenic induction in sSAT. All ASC spheroids increased their capacity to lipolysis under LPS. However, spheroids from dSAT were higher than from sSAT (p = 0.0045) and sRC (p = 0.0005). Newly formed spheroids and spheroids under LPS stimulus from sSAT showed the highest levels of fatty acid-binding protein 4 (FABP4) and CCAAT/enhancer-binding protein-α (C/EBPα) mRNA expression compared with dSAT and sRC (p < 0.0001). ASC spheroids from sRC showed the highest synthesis of angiogenic cytokines such as vascular endothelial growth factor (VEGF) compared with dSAT (p < 0.0228). Under LPS stimulus, ASC spheroids from sRC showed the highest synthesis of pro-inflammatory cytokines such as IL-6 compared with dSAT (p < 0.0092). Conclusion: Distinct physiological properties of SAT can be recapitulated in ASC spheroids. In summary, the ASC spheroid from dSAT showed the greatest lipolytic capacity, from sSAT the greatest adipogenic induction, and sRC showed greater secretory capacity when compared to the dSAT. Together, all these capacities form a true mimicry of SAT and hold the potential to contribute for a deeper understanding of cellular and molecular mechanisms in healthy and unhealthy adipose tissue scenarios or in response to pharmacological interventions.
Keywords: adipogenesis; adipokines; adipose stromal/stem cells; deep subcutaneous adipose tissue; spheroids; subcutaneous adipose tissue; superficial retinacula cutis; superficial subcutaneous adipose tissue.
Copyright © 2023 Côrtes, Alves, Claudio-Da-Silva and Baptista.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Baptista L. S., Côrtes I., Montenegro B., Claudio-da-Silva C., Bouschbacher M., Jobeili L., et al. (2021). A novel conjunctive microenvironment derived from human subcutaneous adipose tissue contributes to physiology of its superficial layer. Stem Cell Res. Ther. 12 (1), 480. 10.1186/s13287-021-02554-9 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

