Purification of linearized template plasmid DNA decreases double-stranded RNA formation during IVT reaction
- PMID: 37842641
- PMCID: PMC10570549
- DOI: 10.3389/fmolb.2023.1248511
Purification of linearized template plasmid DNA decreases double-stranded RNA formation during IVT reaction
Abstract
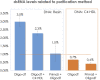
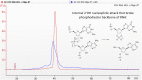
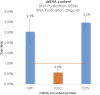
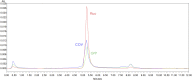
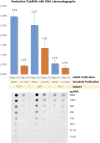
After the COVID-19 pandemic, messenger RNA (mRNA) has revolutionized traditional vaccine manufacturing. With the increasing number of RNA-based therapeutics, valuable new scientific insights into these molecules have emerged. One fascinating area of study is the formation of double-stranded RNA (dsRNA) during in vitro transcription (IVT) which is considered a significant impurity, as it has been identified as a major trigger in the cellular immune response pathway. Therefore, there is a growing importance placed to develop and optimize purification processes for the removal of this by-product. Traditionally, efforts have primarily focused on mRNA purification after IVT through chromatographic separations, with anion exchange and reverse phase chromatography emerging as effective tools for this purpose. However, to the best of our knowledge, the influence and significance of the quality of the linearized plasmid have not been thoroughly investigated. Plasmids production involves the growth of bacterial cultures, bacterial harvesting and lysis, and multiple filtration steps for plasmid DNA purification. The inherent complexity of these molecules, along with the multitude of purification steps involved in their processing, including the subsequent linearization and the less-developed purification techniques for linearized plasmids, often result in inconsistent batches with limited control over by-products such as dsRNA. This study aims to demonstrate how the purification process employed for linearized plasmids can impact the formation of dsRNA. Several techniques for the purification of linearized plasmids based on both, resin filtration and chromatographic separations, have been studied. As a result of that, we have optimized a chromatographic method for purifying linearized plasmids using monolithic columns with C4 chemistry (butyl chains located in the surface of the particles), which has proven successful for mRNAs of various sizes. This chromatographic separation facilitates the generation of homogeneous linearized plasmids, leading to mRNA batches with lower levels of dsRNA during subsequent IVT processes. This finding reveals that dsRNA formation is influenced not only by RNA polymerase and IVT conditions but also by the quality of the linearized template. The results suggest that plasmid impurities may contribute to the production of dsRNA by providing additional templates that can be transcribed into sequences that anneal with the mRNA molecules. This highlights the importance of considering the quality of plasmid purification in relation to dsRNA generation during transcription. Further investigation is needed to fully understand the mechanisms and implications of plasmid-derived dsRNA. This discovery could shift the focus in mRNA vaccine production, placing more emphasis on the purification of linearized plasmids and potentially saving, in some instances, a purification step for mRNA following IVT.
Keywords: IVT; chromatography; dsRNA; immunogenicity; mRNA; polishing; vaccines.
Copyright © 2023 Martínez, Lampaya, Larraga, Magallón and Casabona.
Conflict of interest statement
JM, VL, AL, HM, and DC were employed by the Company Certest Pharma, Certest Biotec.
Figures



Similar articles
-
Understanding the impact of in vitro transcription byproducts and contaminants.Front Mol Biosci. 2024 Jul 10;11:1426129. doi: 10.3389/fmolb.2024.1426129. eCollection 2024. Front Mol Biosci. 2024. PMID: 39050733 Free PMC article. Review.
-
Removing immunogenic double-stranded RNA impurities post in vitro transcription synthesis for mRNA therapeutics production: A review of chromatography strategies.J Chromatogr A. 2025 Jan 11;1740:465576. doi: 10.1016/j.chroma.2024.465576. Epub 2024 Dec 2. J Chromatogr A. 2025. PMID: 39642661 Review.
-
Double-stranded RNA reduction by chaotropic agents during in vitro transcription of messenger RNA.Mol Ther Nucleic Acids. 2022 Aug 4;29:618-624. doi: 10.1016/j.omtn.2022.08.001. eCollection 2022 Sep 13. Mol Ther Nucleic Acids. 2022. PMID: 36090758 Free PMC article.
-
A Facile Method for the Removal of dsRNA Contaminant from In Vitro-Transcribed mRNA.Mol Ther Nucleic Acids. 2019 Apr 15;15:26-35. doi: 10.1016/j.omtn.2019.02.018. Epub 2019 Feb 27. Mol Ther Nucleic Acids. 2019. PMID: 30933724 Free PMC article.
-
Graphene Oxide-Modified Resin for Selective dsRNA Removal from In Vitro-Transcribed mRNA.ACS Appl Bio Mater. 2025 Apr 21;8(4):3541-3551. doi: 10.1021/acsabm.5c00320. Epub 2025 Mar 27. ACS Appl Bio Mater. 2025. PMID: 40150800
Cited by
-
Preclinical evaluation of AGT mRNA replacement therapy for primary hyperoxaluria type I disease.Sci Adv. 2025 Apr 11;11(15):eadt9694. doi: 10.1126/sciadv.adt9694. Epub 2025 Apr 9. Sci Adv. 2025. PMID: 40203111 Free PMC article.
-
Recent Advancements in mRNA Vaccines: From Target Selection to Delivery Systems.Vaccines (Basel). 2024 Aug 1;12(8):873. doi: 10.3390/vaccines12080873. Vaccines (Basel). 2024. PMID: 39203999 Free PMC article. Review.
-
Tuning hydrogen bonds and electrostatics with convection for purifying mRNA: A paradigm shift.Sci Adv. 2025 Jun 20;11(25):eadv8656. doi: 10.1126/sciadv.adv8656. Epub 2025 Jun 18. Sci Adv. 2025. PMID: 40531990 Free PMC article.
-
Effective Synthesis of High-Integrity mRNA Using In Vitro Transcription.Molecules. 2024 May 23;29(11):2461. doi: 10.3390/molecules29112461. Molecules. 2024. PMID: 38893337 Free PMC article.
-
Understanding the impact of in vitro transcription byproducts and contaminants.Front Mol Biosci. 2024 Jul 10;11:1426129. doi: 10.3389/fmolb.2024.1426129. eCollection 2024. Front Mol Biosci. 2024. PMID: 39050733 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

