Washed microbiota transplantation reduces glycemic variability in unstable diabetes
- PMID: 37846600
- PMCID: PMC10859319
- DOI: 10.1111/1753-0407.13485
Washed microbiota transplantation reduces glycemic variability in unstable diabetes
Abstract
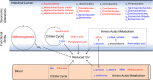
Background: Dysbiosis of gut microbiota is causally linked to impaired host glucose metabolism. We aimed to study effects of the new method of fecal microbiota transplantation, washed microbiota transplantation (WMT), on reducing glycemic variability (GV) in unstable diabetes.
Methods: Fourteen eligible patients received three allogenic WMTs and were followed up at 1 week, 1 month, and 3 months. Primary outcomes were daily insulin dose, glucose excursions during meal tests, and GV indices calculated from continuous monitoring or self-monitoring glucose values. Secondary outcomes were multiomics data, including 16S rRNA gene sequencing, metagenomics, and metabolomics to explore underlying mechanisms.
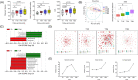
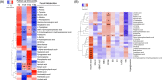
Results: Daily insulin dose and glucose excursions markedly dropped, whereas GV indices significantly improved up to 1 month. WMT increased gut microbial alpha diversity, beta diversity, and network complexity. Taxonomic changes featured lower abundance of genera Bacteroides and Escherichia-Shigella, and higher abundance of genus Prevotella. Metagenomics functional annotations revealed enrichment of distinct microbial metabolic pathways, including methane biosynthesis, citrate cycle, amino acid degradation, and butyrate production. Derived metabolites correlated significantly with improved GV indices. WMT did not change circulating inflammatory cytokines, enteroendocrine hormones, or C-peptide.
Conclusions: WMT showed strong ameliorating effect on GV, raising the possibility of targeting gut microbiota as an effective regimen to reduce GV in diabetes.
Keywords: fecal microbiota transplantation; glycemic variability; hypoglycemia; microbiome; unstable diabetes.
© 2023 The Authors. Journal of Diabetes published by Ruijin Hospital, Shanghai Jiaotong University School of Medicine and John Wiley & Sons Australia, Ltd.
Figures



References
-
- Smith‐Palmer J, Brandle M, Trevisan R, Orsini Federici M, Liabat S, Valentine W. Assessment of the association between glycemic variability and diabetes‐related complications in type 1 and type 2 diabetes. Diabetes Res Clin Pract. 2014;105:273‐284. - PubMed
-
- Jin SM, Kim TH, Bae JC, et al. Clinical factors associated with absolute and relative measures of glycemic variability determined by continuous glucose monitoring: an analysis of 480 subjects. Diabetes Res Clin Pract. 2014;104:266‐272. - PubMed
-
- Brownlee M, Hirsch IB. Glycemic variability: a hemoglobin A1c‐independent risk factor for diabetic complications. JAMA. 2006;295:1707‐1708. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

