Design and Optimization of Novel Competitive, Non-peptidic, SARS-CoV-2 Mpro Inhibitors
- PMID: 37849558
- PMCID: PMC10577883
- DOI: 10.1021/acsmedchemlett.3c00335
Design and Optimization of Novel Competitive, Non-peptidic, SARS-CoV-2 Mpro Inhibitors
Abstract
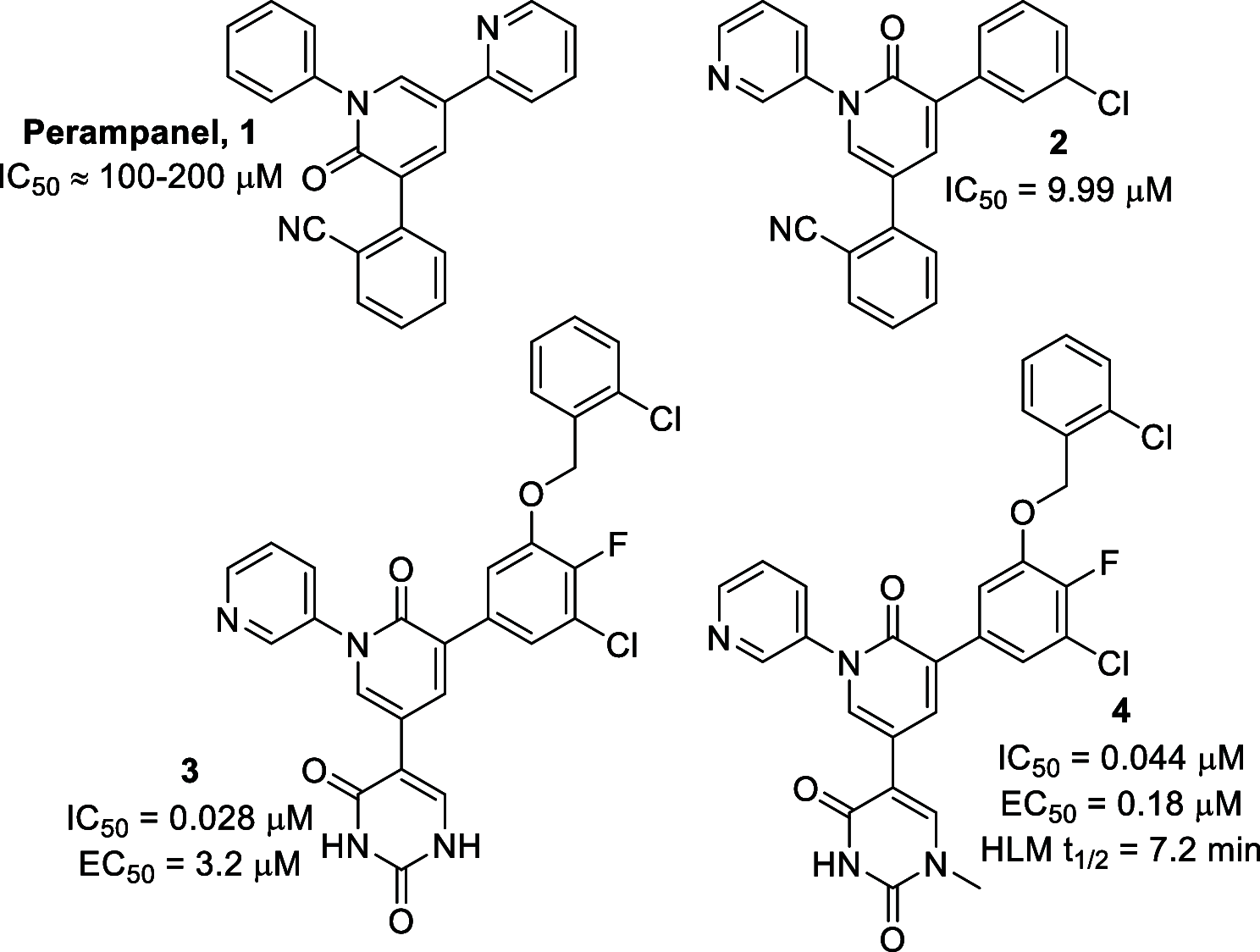
The SARS-CoV-2 main protease (Mpro) has been proven to be a highly effective target for therapeutic intervention, yet only one drug currently holds FDA approval status for this target. We were inspired by a series of publications emanating from the Jorgensen and Anderson groups describing the design of potent, non-peptidic, competitive SARS-CoV-2 Mpro inhibitors, and we saw an opportunity to make several design modifications to improve the overall pharmacokinetic profile of these compounds without losing potency. To this end, we created a focused virtual library using reaction-based enumeration tools in the Schrödinger suite. These compounds were docked into the Mpro active site and subsequently prioritized for synthesis based upon relative binding affinity values calculated by FEP+. Fourteen compounds were selected, synthesized, and evaluated both biochemically and in cell culture. Several of the synthesized compounds proved to be potent, competitive Mpro inhibitors with improved metabolic stability profiles.
© 2023 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures






References
-
- Wu F.; Zhao S.; Yu B.; Chen Y.-M.; Wang W.; Song Z.-G.; Hu Y.; Tao Z.-W.; Tian J.-H.; Pei Y.-Y.; Yuan M.-L.; Zhang Y.-L.; Dai F.-H.; Liu Y.; Wang Q.-M.; Zheng J.-J.; Xu L.; Holmes E. C.; Zhang Y.-Z. A new coronavirus associated with human respiratory disease in China. Nature 2020, 579 (7798), 265–269. 10.1038/s41586-020-2008-3. - DOI - PMC - PubMed
-
- Andrews N.; Stowe J.; Kirsebom F.; Toffa S.; Rickeard T.; Gallagher E.; Gower C.; Kall M.; Groves N.; O’Connell A.-M.; Simons D.; Blomquist P. B.; Zaidi A.; Nash S.; Iwani Binti Abdul Aziz N.; Thelwall S.; Dabrera G.; Myers R.; Amirthalingam G.; Gharbia S.; Barrett J. C.; Elson R.; Ladhani S. N.; Ferguson N.; Zambon M.; Campbell C. N. J.; Brown K.; Hopkins S.; Chand M.; Ramsay M.; Lopez Bernal J. Covid-19 Vaccine Effectiveness against the Omicron (B.1.1.529) Variant. N. Engl. J. Med. 2022, 386 (16), 1532–1546. 10.1056/NEJMoa2119451. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Chemical Information
Miscellaneous

