The use of nanomaterials in advancing photodynamic therapy (PDT) for deep-seated tumors and synergy with radiotherapy
- PMID: 37849983
- PMCID: PMC10577272
- DOI: 10.3389/fbioe.2023.1250804
The use of nanomaterials in advancing photodynamic therapy (PDT) for deep-seated tumors and synergy with radiotherapy
Abstract
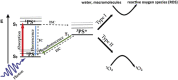
Photodynamic therapy (PDT) has been under development for at least 40 years. Multiple studies have demonstrated significant anti-tumor efficacy with limited toxicity concerns. PDT was expected to become a major new therapeutic option in treating localized cancer. However, despite a shifting focus in oncology to aggressive local therapies, PDT has not to date gained widespread acceptance as a standard-of-care option. A major factor is the technical challenge of treating deep-seated and large tumors, due to the limited penetration and variability of the activating light in tissue. Poor tumor selectivity of PDT sensitizers has been problematic for many applications. Attempts to mitigate these limitations with the use of multiple interstitial fiberoptic catheters to deliver the light, new generations of photosensitizer with longer-wavelength activation, oxygen independence and better tumor specificity, as well as improved dosimetry and treatment planning are starting to show encouraging results. Nanomaterials used either as photosensitizers per se or to improve delivery of molecular photosensitizers is an emerging area of research. PDT can also benefit radiotherapy patients due to its complementary and potentially synergistic mechanisms-of-action, ability to treat radioresistant tumors and upregulation of anti-tumoral immune effects. Furthermore, recent advances may allow ionizing radiation energy, including high-energy X-rays, to replace external light sources, opening a novel therapeutic strategy (radioPDT), which is facilitated by novel nanomaterials. This may provide the best of both worlds by combining the precise targeting and treatment depth/volume capabilities of radiation therapy with the high therapeutic index and biological advantages of PDT, without increasing toxicities. Achieving this, however, will require novel agents, primarily developed with nanomaterials. This is under active investigation by many research groups using different approaches.
Keywords: X-ray PDT; XPDT; immune activation; photodynamic therapy (PDT); radiation; radioPDT.
Copyright © 2023 Dinakaran and Wilson.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Almahwasi A. (2016). Does hadron therapy offer enough effectiveness in treating cancer to be worth the cost?
Publication types
LinkOut - more resources
Full Text Sources

