Real-world treatment patterns of rheumatoid arthritis in Brazil: analysis of DATASUS national administrative claims data for pharmacoepidemiology studies (2010-2020)
- PMID: 37853013
- PMCID: PMC10584810
- DOI: 10.1038/s41598-023-44389-9
Real-world treatment patterns of rheumatoid arthritis in Brazil: analysis of DATASUS national administrative claims data for pharmacoepidemiology studies (2010-2020)
Abstract
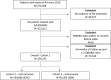
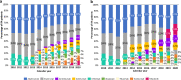
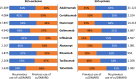
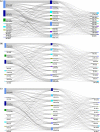
Our study assessed DATASUS as a potential source for pharmacoepidemiologic studies in rheumatoid arthritis (RA) in the Brazilian population focusing on treatment patterns and determinants of initiating or switching to a novel therapy. This was a descriptive database study of RA patients with at least one claim of RA and ≥ 2 claims of disease-modifying anti-rheumatic drug (DMARD); conventional synthetic (cs), biologic (b) or targeted synthetic (ts) DMARD with more than 6 months of follow-up from 01-Jan-2010 to 31-Dec-2020. Analyses were stratified for SUS-exclusive and SUS+ private user cohorts. We identified 250,251 patients with RA in DATASUS: mean age of 58.4 years, majority female (83%) and white (58%). 62% were SUS-exclusive and 38% SUS+ private. Most common bDMARDs were adalimumab and etanercept. Age (adjusted odds ratio 1.78 [50+]; 95% CI 1.57-2.01), SUS exclusive status (0.53; 0.47-0.59), distance to clinic [160+ km] (0.57; 0.45-0.72), and pre-index csDMARD claims (1.23; 1.08-1.41) were independent predictors of initiating a novel oral tsDMARD. Switching from bDMARD to tsDMARD, associations were similar, except for the direction of associations for SUS exclusive status (adjusted hazard ratio 1.10; 1.03-1.18), distance to clinic (1.18; 1.03-1.35), and number of previous bDMARD (0.15; 0.14-0.16). DATASUS is a source suitable for treatment-related analyses in RA reflecting the public health system in Brazil.
© 2023. Springer Nature Limited.
Conflict of interest statement
Competing interests are financial in nature and associated with employment. MGB was an employee of IQVIA (Brazil) at the time of study conduct, and currently has no conflict of interest to declare. RF is an employee of IQVIA. WSK, MC, and AL are employees and stockholders with AbbVie Inc. GSJ was an employee of IQVIA (Brazil) at the time of study conduct, and is currently an employee and stockholder with Pfizer (Brazil). Otherwise, authors do not have further non-financial competing interests to declare.
Figures
References
-
- U.S. Food and Drug. Real-World Evidence. https://www.fda.gov/science-research/science-and-research-special-topics... (2023).
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

