Retinoic Acid Signaling in Fatty Liver Disease
- PMID: 37854944
- PMCID: PMC10583737
- DOI: 10.1016/j.livres.2023.07.002
Retinoic Acid Signaling in Fatty Liver Disease
Abstract
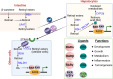
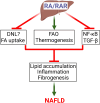
Retinoic acid (RA) is a metabolite of vitamin A and is essential for development and growth as well as cellular metabolism. Through genomic and nongenomic actions, RA regulates a variety of physiological functions. Dysregulation of RA signaling is associated with many diseases. Targeting RA signaling has been proven valuable to human health. All-trans retinoic acid (AtRA) and anthracycline-based chemotherapy are the standard treatment of acute promyelocytic leukemia (APL). Both human and animal studies have shown a significant relationship between RA signaling and the development and progression of nonalcoholic fatty liver disease (NAFLD). In this review article, we will first summarize vitamin A metabolism and then focus on the role of RA signaling in NAFLD. AtRA inhibits the development and progression of NAFLD via regulating lipid metabolism, inflammation, thermogenesis, etc.
Keywords: Retinoic acid; fatty acid oxidation; fatty liver disease; lipogenesis; obesity.
Conflict of interest statement
The authors declare that there is no conflicts of interest.
Figures
References
-
- Bashir A., Duseja A., De A., Mehta M., Tiwari P. Non-alcoholic fatty liver disease development: a multifactorial pathogenic phenomena. Liver Res. 2022;6:72–83. doi: 10.1016/j.livres.2022.05.002. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
